The structure of matter has been a subject of speculation from very early times. According to greek philosopher Aemocritus, if we
go on dividing matter into smaller parts, a stage would be reached when particles obtained cannot be divided further. He called these
particles 'atoms' meaning indivisible.
Conclusion : All matter is made up of small particles called atoms. Different kinds of atoms and molecules have different properties
due to which different kinds of matter also show different properties.
By studying the result of quantitative measurement of many reactions it was observed that whenever substances react, they follow certain
laws. These laws are called the law of chemical combination.
(a) Law of conservation of mass. (b) Law of constant proportions. (c) Law of multiple proportions.
(a) Law of conservation of mass ;
This law was given by the french chemist A. Lavoisier in 1774. This law states that in every chemical reaction, the total mass before and
after the reaction remains constant.
"That is mass can neither be created nor destroyed in a chemical reaction". Lavoisier showed that when mercuric oxide was heated, it produced
free mercury and oxygen. The sum of masses of mercury and oxygen was found to be equal to the mass of mercuric oxide.
Mercuric oxide ® Mercury + Oxygen
100 g 92.69 g 7.49 g
Activity : Demonstration of law of conservation of mass.
* Prepare separately a 5% solution of barium chloride and a 5%
solution of sodium sulphate.
* Take about 20ml of barium chloride solution in a conical flask.
* Take sodium sulphate solution in a small test tube. Hang the
test tube in the conical flask with the help of a thread.
Close the mouth of the flask with cork.
* Weigh the flask along with its contents.
* Now tilt the flask so that the two solution get mixed.
* Weigh the flask again along with its contents. What do you observe ? It is observed that on mixing the two solution a chemical reaction takes
place which is indicated by the formation of a white precipitate
Barium chloride + Sodium sulphate ® Barium sulphate (white ppt) + Sodium Chloride
* The mass of the flask and its contents remains constant. Thus, during a chemical reaction mass is neither created nor destroyed.
This activity can also be carried out with the following pairs :
(a) Silver nitrate and sodium chloride (b) Copper sulphate and sodium carbonate
Q. In a reaction 5.3 g of sodium carbonate reacted with 6 g of ethanoic acid. The products were 2.2 g of carbon dioxide. 0.9 g water and 8.2 g of
sodium ethanoate. Show that these observations are in agreement with the law of conservation of mass. [NCERT]
Sodium carbonate + ethanoic acid ® sodium ethanoate + carbon dioxide + water.
Q. Hydrogen and oxygen combine in the ratio of 1 : 8 by mass to form water. What mass of oxygen gas would be required to react completely with 3 g of hydrogen gas? [NCERT]
Q. Which postulate of Dalton's atomic theory is the result of the law of conservation of mass? [NCERT]
(b) Law of constant Proportions / Law of definite proportions :
This law was given by the french chemist A. Lavoisier and Joseph Proust. This law deals with the composition of chemical compounds.
This law is : A pure chemical compound always contains same elements combined together in same proportion by mass.
For example : Pure water obtained from different sources such as river, well etc. always contains hydrogen and oxygen
combined together in the ratio 1 : 8 by mass similarly carbon dioxide can be obtained by different methods such as by burning of
carbon, by heating lime stone. It shows that samples of carbon dioxide obtained from different sources contain carbon and oxygen in
the ratio of 3 : 8 by mass. Thus in water or CO2 this proportion of hydrogen and oxygen or carbon and oxygen always remains constant.
Ex. Hydrogen and oxygen combine in the ratio of 1 : 8 by mass to form water. What weight of oxygen gas would be required to completely react with
3 g of hydrogen gas.
Sol. Ratio in which hydrogen and oxygen combine = 1 : 8
1 g of hydrogen combines with oxygen = 8 g
\ 3 g of hydrogen will combine with oxygen = 8 × 3 = 24 g.
(c) Law of multiple proportions :
It was given by Dalton in 1808. According to it, when one element combines with the other element to from two or more different compounds,
the mass of one element, which combines with a constant mass of the other, bear a simple ratio to one another. Example : Carbon and oxygen
when combine, can from two oxides that are CO (carbon monoxide), CO2 (carbon dioxide).
In CO, 12g carbon combine with 16g of oxygen. In CO2, 12g carbon combine with 32g of oxygen. Thus, we can see the mass of
oxygen which combine with a constant mass of carbon (12g) bear simple ratio of 16 : 32 or 1 : 2.
On the basis of laws of chemical combination John Dalton proposed atomic theory in 1808. The main points of dalton's atomic theory are :
The main postulates of the Dalton's atomic theory are :
1. All matter is made up of very tiny particles called atoms.
2. Atoms are indivisible particles, which cannot be created or destroyed in a chemical reaction,
3. Atoms of a given element are identical in mass size and chemical properties.
4. Atoms of different element have different mass size and chemical properties.
5. Atoms combine in the ratio of small whole numbers to form compounds.
6. The relative number and kinds of atoms are constant in a given compound.
Dalton's atomic theory was based on the laws of chemical combination.
7. Atoms of same element can combine in more than one ratio to form more than one compound. For example, hydrogen and oxygen combine to
give water and hydrogen peroxide. In water, two atoms of hydrogen combine with one atom of oxygen while in hydrogen peroxide, two
atoms of hydrogen combine with two atoms of oxygen.
For example : The postulates of Dalton's atomic theory that "atoms can neither be created nor destroyed", was the result of law of
conservation of mass and the postulates of Dalton's atomic theory that "the element consist of atom having fixed mass", and
that the number and kind of atom in a given compound is fixed came from the law of constant proportions.
Q. Which postulate of Dalton's atomic theory can explain the law of definite proportions? [ncert]
Drawbacks of Dalton's Atomic Theory :
Some of the drawbacks of the Dalton's atomic theory of matter are given below :
l According to Dalton's atomic theory, atoms were thought to be indivisible. But it is now known that atoms can be further divided into
still smaller particle called electrons, protons and neutrons.
l Dalton's atomic theory said that all the atoms of an element have exactly the same mass. But it is now known that atoms of the same
element can have slightly different masses, as in case of isotopes.
l Dalton's atomic theory said that atoms of different elements have different masses. But it is now known that even atoms of different
elements can have the same mass as in case of isobars.
all the matter is made up of atoms. An atom is the smallest particles of an element that can take part in a chemical reaction. Atoms of most of
the elements are very reactive and do not exist in the free state
(as single atom). They exist in combination with the atoms of the same elements or another element. Atoms are very small in size.
The size of an atom is indicated by its radius which is called atomic radius (radius of an atom). Atomic radius is measured in nanometre (nm)
(1 metre = 109 nanometres or 1nm =
10–9m). Hydrogen atoms is the smallest atom of all, having an atomic radius of 0.037nm. Atoms are so small that we cannot see them under the
most powerful optical microscope.
Symbol may be defined as the abbreviation used for the name of an element. The symbol of an element are generally either the first letter
or the first two letters or the first and the third letters of the name of the element. for example, the symbol of the following elements are
the first letter of the name of that element.
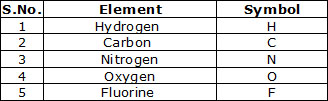
l Some symbols derived from the first two letters of the names of the element.
2 Some symbol derived from the first and the third letter of the names of the elements.
3 There are certain symbols which seen to here no relationship to their names. The symbol of these elements are derived from their latin names.
Atomic Mass : Atomic mass of an element may be defined as the average relative mass of an atom of the element as compared with mass
of an atom of carbon (C-12 isotope) taken as 12 amu.
Atomic mass :
How do Atoms occur :
The atoms of only a few element called noble gases (such as helium, neon, argon and krypton etc.) which are chemically unreactive and exist
in the free state (as single atom). Atoms of the elements are chemically very reactive and do not exist in the free state (as single atom)
Atoms usually exist in two ways :
Q. Define the atomic mass unit. [NCERT]
Q. Why is it not possible to see an atom with naked eyes? [NCERT]
A molecule is the smallest particle of an element or compound that has independent existence. A molecule contain one or more than one atoms.
The molecules of elements contain atoms of only one kind.
The number of atoms in a molecule of an element is known as atomicity of the element. For example -The atomicity of the noble gases is 1, that of
hydrogen, nitrogen, oxygen etc. is 2 each and of ozone is 3. Thus, noble gases, hydrogen and ozone are respectively monoatomic, diatomic and triatomic molecules.
Molecules of elements :
The molecules of an element contain two similar atom chemically bonded together, for example ozone gas has 3 oxygen atoms combined together ,
so ozone exists in the form of O3. A recently discovered form of carbon, called Buckminster fullerene has molecular formula C60.
Molecules of compounds :
The molecules of a compound contain two or more different types of atoms chemically bonded together. For example : the molecule sulphur
dioxide (SO2) contain one atom of sulphur chemically bonded with two atom of oxygen.
Molecular mass and formula mass :
The molecular mass of a substance (an element or a compound) may be defined as the average relative mass of a molecule of the substance as
compared with mass of an atom of carbon (C-12 isotope) taken as 12 amu.
Molecular Mass =
The molecular mass of a compound can be obtained by adding atomic masses of all the atoms present in the molecule of the compound. For example,
molecular mass of CO2 is -
12 × 1 + 16 × 2 = 44 u
Gram Molecular Mass :
Gram molecular mass of a substance is defined as that much quantity of the substance whose mass expressed in grams is numerically equal to its molecular mass.
For example : The molecular mass of CO2 is 44 u, its gram molecular mass is 44g. Gram molecule mass of a substance is also known as gram-molecular mass of the substance.
Formula Mass :
Formula mass of an ionic compound is obtained by adding atomic masses of all the atoms in a formula unit of the compound.
For example : Formula mass of potassium chloride (KCl)
= Atomic mass of potassium + atomic mass of chlorine
39 + 35.5 = 74.5
Q. Calculate the molecular masses of H2, O2, Cl2, CO2, CH4, C2H6, C2H4, NH3, CH3OH. [ncert]
Q. Calculate the formula unit masses of ZnO, Na2O, K2CO3, given atomic masses of Zn = 65 u, Na = 23 u,
K = 39 u, C = 12 u and O = 16 u [ncert]
Q. Calculate the molar mass of the following substances. [ncert]
(a) Ethyne, C2H2
(b) Sulphur molecule, S8
(c) Phosphorous molecule, P4(Atomic mass of phosphorus = 31)
(d) Hydrochloric acid, HCl
(e) Nitric acid, HNO3
Chemical formula :
The chemical formula of a compound describes the composition of a molecule of the compound in terms of the symbols of elements and the
number of atoms of each element present in one molecule of the compound.
l In the chemical formula of a compound, the elements present are denoted by their symbols and the number of atoms of each element
are denoted by writing their number as subscripts to the symbols of the respective element.
Example : Water is a compound whose one molecule is made up of two atoms of hydrogen and one atom of oxygen and hence its chemical formula is H2O.
l While writing the formula of an ionic compound the metal is written on the left hand side while the non-metal is written on the right h
and side. The name of the metal remains as such but that of the non-metal is changed to have the ending 'ide'.
Example : MgO is named as magnesium oxide, KCl is named potassium chloride etc.
l Molecular compounds, formed by the combination between two different non-metals, are written in such a way that the less electronegative
element is written on the left hand side while the more electronegative element is written on the right hand side. In naming molecular
compounds, the name of the less negative non-metal is written as such but the name of the more electronegative element is changed to have the ending 'ide'.
Example : H2S is named as hydrogen sulphide.
l When there are more than one atoms of an element are present in the formula of the compound, then the number of atoms are indicated by
the use of appropriate prefixes (Mono for : 1, di for 2, tri for 3. tetra for 4 atoms etc.) in the name of the compound.
Example : CO2 is named as carbon di oxide, CCl4 is named as carbon tetra chloride.
The prefixes are needed in naming those binary compounds in which the two non-metals form more than one compounds (by having different number of atoms).
Example : Two non-metal, nitrogen and oxygen, combine to form different compound like nitrogen monoxide (NO), nitrogen di-oxide (NO2), Nitrogen tri oxide (N2O3) etc.
l But, if two non-metals form only one compound, then prefixes are not used in naming such compounds.
Example : Hydrogen and sulphur combine to form only one compound H2S, So, H2S is named as hydrogen sulphide and not hydrogen monosulphide.
Ions : An ion is a positively or negatively charged atom (or group of atoms)
These are two type of ions :
(1) cations (2) anions
(1) Cations : A positively charged ion is known as cation. For example : Sodium ion:Na+, Magnesium ion: Mg2+
A cation is formed by the loss of one or more electrons by an atom
For example : sodium atom, loses one electron to form a sodium ion Na+
Na Na+ sodium ion
Sodium atom (A cation)
(2) Anions : A negatively charged ion is known as anion. Cl– (chloride ion), O–2 (oxide ion) etc.
An anions is formed by the gain of one or more electrons by an atom. For example a chlorine atom gains one electron to form a chloride ion Cl–.
l Cl Cl–
Chlorine atom Chloride ion (An anion)
Valency of ions : The valency of an ion is same as the Charge present on the ion.
* Monovalent cation (Valency of cation +1)
Example : Sodium ion (Na+). Potassium ion (K+), Hydrogen ion (H+).
* Divalent cations (valency of cations + 2)
Example : Magnesium ion (Mg+2) Ferrous ion (Fe+2)
* Trivalent cations (valency of cations + 3)
Example : Aluminium ion (Al+3), Ferric ion (Fe+3).
* Monovalent anion (anion of valency –1)
Example : Chloride ion (Cl–), Bromide ion (Br–)
* Divalent anions (Anions of Valency –2)
Example : Oxide ion (O–2), Peroxide ion (O2–2) etc.
Trivalent anion (anions of valency –3)
Example : Nitride ion (N–3), Phosphate ion (PO4–3) etc.
Writing of formula of Molecular compound :
Steps : . The steps to be followed for writing the formula of molecular compound are-
l First, write the symbols of the elements contributing in the compound.
2 Then, below each symbol, write its corresponding valency
3 Finally, we exchange the valencies of the combining atoms that is with first atom, we write the valency of the second atom and with second atom,
we write the valency of the first atom, the valencies to be written as subscripts to the symbols.
4 If the valencies have any common factor, then the formula is divided by the common factor. This gives the required formula of the compound.
Example : To work out the formula of hydrogen sulphide
(1) Hydrogen sulphide compound is made up of hydrogen and sulphur elements. So first we write down the symbol of hydrogen and sulphur.
(2) The valency of hydrogen is 1 and the valency of sulphur is 2. So below the symbol H we write 1 and below the symbol S we write 2.

5 We now cross-over the valencies of H and S atoms. With H atom we write the valency of S (which is 2) so that it becomes H2 with S atom we write the valency of H
(which is 1) so that it becomes S1. Now, joining together H2 and S1 the formula of hydrogen sulphide becomes H2S1 or H2S
(This is because we don't write the subscript 1 with an atom in a formula).
Q. What is meant by the term chemical formula? [ncert]
Q. How many atoms are present in a [ncert]
(i) H2S molecule and (ii) PO43– ion?
Q. What are polyatomic ions? Give examples [ncert]
Q. Give the names of the elements present in the following compounds. [ncert]
(a) Quick lime (b) Hydrogen bromide (c) Baking powder (d) Potassium sulphate
Example : To write the formula for sodium carbonate.
(1) First, write the symbol of sodium ion and carbonate ion and write their valencies below their symbols are shown.
Symbols Na CO3
Valencies +1 –2
(or charges)
(2) Now, we exchange the valencies of sodium ion and carbonate ion,
(3) So –2 gets associated with Na and +1 gets associated with CO3 in this way we get Na2 and CO3 and final formula of sodium carbonate is Na2CO3.
Q. Write down the formulae of [ncert]
(i) sodium oxide (ii) aluminium chloride (iii) sodium sulphide (iv) magnesium hydroxide
Q. Write down the names of compounds represented by the following formulae : [ncert]
(i) Al2(SO4)3 (ii) CaCl2 (iii) K2SO4
(iv) KNO3 (v) CaCO3
Q. Write the chemical formulae of the following. [ncert]
(a) Magnesium chloride (b) Calcium oxide (c) Copper nitrate
(d) Aluminium chloride (e) Calcium carbonate
– 1 Charge – 2 Charge – 3 Charge
Fluoride F– Sulphide S2– Phosphide P3–
Chloride Cl– Oxide O2– Nitride N3–
Bromide Br–
Iodide I–
Hydride H–
Mole : Mole is a link between the mass of atoms (or molecules) and the number of atoms (or molecules). A group of 6.022 × 1023 particles
(atom, molecules or ions) of a substance is called a mole of that substance.
Thus , 1 mole of atoms = 6.023 × 1023 atoms.
1 mole of molecules = 6.022 × 1023 molecules.
For example : oxygen atom is O and oxygen molecule is O2.
1 mole of oxygen atoms (O) = 6.022 × 1023 oxygen atom
1 mole of oxygen molecules = 6.022 × 1023 oxygen molecules.
Number of 6.022 × 1023, which represents a mole is known as Avogadro number.
Moles of Atoms : One mole of atoms of an element has a mass equal to the gram atomic mass of the element.
1 mole of atoms of an element = Gram atomic mass of the element.
For example : The atomic mass of oxygen (O) is 16 u, so gram atomic mass of oxygen will be 16 gram.
1 mole of oxygen atoms = Gram atomic mass of oxygen = 16 gram.
Mole of Molecules :
1 mole of molecules of an substance has mass equal to the gram molecular mass of the substance.
1 mole of molecules of a substance = Gram molecular mass of the substance.
For example : The molecular mass of oxygen (O2) is 32 u. So the gram molecular mass of oxygen molecule is 32 grams.
1 mole of oxygen molecules = Gram molecular mass of oxygen = 32 gram.
The molecular formula of a compound may be defined as the formula which specifies the number of atoms of various element in the molecule of the compound.
For example : The molecular formula of glucose is C6H12O6. This shows that a molecule of glucose contains six atoms of carbon, twelve atoms of hydrogen and six atoms of oxygen. With the help of molecular formula of a compound we can calculate its percentage composition by mass. First we calculate the molecular mass of the compound. From this we can find out mass of one mole of the compound, which is equal to its gram molecular mass. Then we calculate mass of each element in one mole of the compound. The mass percentage of each element is then calculated by the following formula.
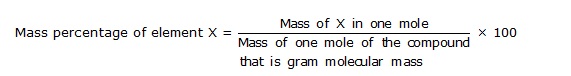 × 100
× 100 To find out the molecular formula of a compound, the first is to determine its empirical formula from the percentage composition. The empirical
formula of a compound may be defined as the formula which gives the simplest whole number ratio of atoms of the various elements present in the
molecule of the compound.
For example : The empirical formula of the compound glucose (C6H12O6) is CH2O which shows that C, H and O are present in the simplest ratio of 1 : 2 : 1.
Molecular formula is whole number multiple of empirical formula thus,
Molecular formula = Empirical formula × n

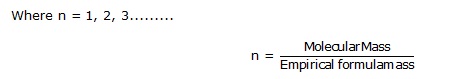
The percentage of the element in the compound is determined by suitable methods and from the data collected, the empirical formula is determined by the following steps-
While doing numerical problems of mole concept, the following relations should be remembered.
Key for symbols
Number of moles = n
Given mass = m
Molar mass = M
Given number of particle = N
Avogadro number of particle = N0
Key Formula
1 mole of atoms = 6.022 × 1023 atoms = Gram atomic mass or Molar mass of element
Number of moles = Mass of element/Molar mass
n= m/M
Number of moles = Given number of atoms/Avogadro number
These relations can be interchanged as
Mass of element, m = n × M
or Number of particles of element, N = n × N0
Similarly,
1 mole of molecules = 6.022 × 1023 molecules = Gram molecular mass or Molar mass
Number of moles = Mass of substance/Molar mass
n= m/M
Number of moles = Given number of molecules / Avogadro number
or m = n × M and N = n × N0
Let us apply these formula to solve numerical problems.
¨ An element is a ure substance which cannot be split into two or more simpler substance s by any physical or chemical means. It is the simplest form of matter.
¨ Elements may be metals, nonmetals or metalloids.
Q.1 In a reaction, 5.3 g of sodium carbonate reacted with 6 g of ethanoic acid. The products were 2.2 g of carbon dioxide, 0.9 g
water and 8.2 g of sodium ethanoate. Show that these observations are in agreement with the law of conservation of mass.
Sodium carbonate + Ethanoic acid ¾® Sodium Ethanoate + Carbon dioxide + Water
Ans. Sodium carbonate + Ethanoic acid ¾® Sodium Ethanoate + Carbon dioxide + Water
(5.3g) (6g) (8.2g) (2.2g) (0.9g)
Mass of reactants = Mass of sodium carbonate + Mass of ethanoic acid
= 5.3 + 6 = 11.3g
Mass of products = Mass of sodium ethanoate + Mass of carbon dioxide + Mass of water
= 8.2 + 2.2 + 0.9 = 11.3g
Here, mass of reactants = mass of products. It means there is no net change in the mass, i.e., before the reaction and after the
reactions, i.e., the mass is constant. So, these observations are in
agreement with the law of conservation of mass.
Q.2 Hydrogen and oxygen combine in the ratio of 1:8 by mass to form water. What mass of oxygen gas would be required to react completely with 3g of hydrogen gas?
Ans. 1 g hydrogen requires 8 g of oxygen.
\ 3g of hydrogen requires = 3 × 8 = 24 g of oxygen.
Q.3 Which postulate of Dalton’s atomic theory is the result of the law of conservation of mass?
Ans. Atoms are indivisible which can neither be created nor destroyed in a chemical reaction. this is the postulate which is based on the law of conservation of mass.
Q.4 Which postulate of Dalton’s atomic theory can explain the law of definite proportions?
Ans. “The relative number and kind of atoms are constant in a given compound.” this is the postulate which explains the law of definite proportion.
Q.6 Define the atomic mass unit.
Ans. The atomic mass unit (amu) is equal to 1/12th part of the mass of one atom of C-12.
1 amu = 1/12th part of C-12 isotope of carbon.
Q.7 Why is it not possible to see an atom with naked eyes?
Ans. Because the size of atom is very small which is beyond the capacity of our eyes, as the atoms are in the range of 10–9 – 10–10 m.
Q.8 Write down the formulae of
(i) sodium oxide (ii) aluminium chloride
(iii) sodium sulphide (iv) magnesium hydroxide
Ans. (i) sodium oxide = Na2O (ii) Aluminium chloride = AlCl3
(iii) Sodium sulphide = Na2S (iv) Magnesium hydroxide = Mg(OH)2
Q.9 Write down the names of compounds represented by the following formulae:
(i) Al2(SO4)3 (ii) CaCl2 (iii) K2SO4 (iv) KNO3 (v) CaCO3
Ans. (i) Al2(SO4)2 = Aluminium sulphate (ii) CaCl2 = Calcium chloride
(iii) K2SO4 = Potassium sulphate (iv) KNO3 = Potassium nitrate
(v) CaCO3 = Calcium carbonate
Q.10 What is meant by the term chemical formula?
Ans. The short hand notation or representation of a compound or element is called its chemical formula. It represents the composition of a molecule of
the substance in terms of the symbols of the elements present in the molecule. For example, ammonia is a compound and its chemical formula is NH3.
Q.11 How many atoms are present in a:
(i) H2S molecule (ii) PO43– ion?
Ans. (i) H2S molecule = 3 atoms (ii) PO43– ion = 5 atoms
Q.12 Calculate the molecular masses of H2, O2, Cl2, CH4, C2H6, C2H4, NH3, CH3OH.
Ans. H2 = 2 × H O2 = 2 × O Cl2 = 2 × Cl
= 2 × 1 = 2 × 16 = 2 × 35.5
= 2 u = 32 u = 71 u
CO2 = 1 × C + 2 × O CH4 = 1 × C + 4 × H C2H6 = 2 × C + 6 × H
= 1 × 12 + 2 × 16 = 12 + 4 = 2 × 12 + 6 × 1
= 44 u = 16 u = 30 u
C2H4 = 2 × C + 4 × H NH3 = 1 × N + 3 × H CH3OH = 1 × C + 4 × H + 1 × O
= 24 + 4 = 1 × 14 + 3 × 1 = 1 × 12 + 4 × 1 + 1 × 16
= 28 u = 17 u = 12 + 4 + 16
= 32 u
Q.13 Calculate the formula unit masses of ZnO, Na2O, K2CO3, given atomic masses of
Zn = 65u, Na = 23u, K = 39u, C = 12u, O = 16u.
Ans. Formula unit mass of
ZnO = 1 × Zn + 1 × O Na2O = 2 × Na + 1 × O K2CO3 = 2 × K + 1 × C + 3 × O
= 1 × 65 + 1 × 16 = 2 × 23 + 1 × 16 = 2 × 39 + 1 × 12 + 3 × 16
= 81 u = 46 + 16 = 78 + 12 + 48
= 62 u = 138 u
Q.14 If one mole of carbon atoms weighs 12g. What is the mass (in grams) of 1 atom of carbon?
Ans. We know that one mole = 6.022 × 1023 atoms
The mass of 6.022 × 1023 atoms = 12g
\ The mass of one atom = g = 1.99 × 10–23 g
Q.15 Which has more number of atoms: 100g of sodium or 100g of iron (given atomic mass of Na = 23u,
Fe = 56u)?
Ans. To know the number of atoms, we must know the number of moles, (n) =
Number of moles of Na = mole
Number of atoms of Na = n × N0 (N0 = 6.022 × 1023).
= = 2.62 × 1024 atoms
Number of atoms of Fe =
= 1.075 × 1024 atoms
Therefore, 100 grams of Na will have more number of atoms.
Q.16 A 0.24g sample of compound of oxygen and boron was found by analysis to contain 0.096 g of boron and 0.144 g of oxygen. Calculate the percentage composition of the compound by weight.
Ans. % of the element =
% of boron = = = = 40%
% of oxygen = = = = 60%
Q.17 When 3.0g of carbon is burnt in 8.00 g of oxygen, 11.00 g of carbon dioxide is produced. What mass of carbon dioxide will be formed when 3.00 g of carbon
is burnt in 50.00 g of oxygen. Which law of chemical combination will govern your answer?
Ans. C + O2 ¾® CO2
3g 8g 11g
Mass of reactants = Mass of carbon + Mass of oxygen = 3 + 8 = 11g, Mass of products (CO2) = 11g
\ Mass of reactants = Mass of products
So, here the law of conservation of mass is followed. If 3 grams of carbon burns in 50g of oxygen even then it will produce 11g of CO2 because 3 grams of carbon will
combine with fixed amount of oxygen, i.e., 8g. So, the law of constant composition is also followed.
Q.18 What are polyatomic ions? Give examples.
Ans. Those ions which have more than two atoms (group of atoms) are called polyatomic ions.
Example: CO32– (carbonate ion), SO42– (sulphate ion)
PO43– (phosphate ion), NH4+ (ammonium ion)
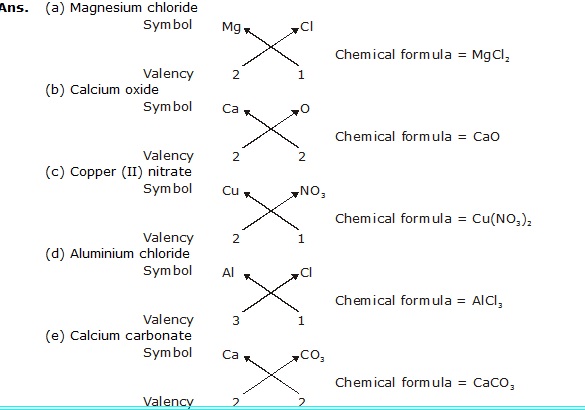
Q.19 Write chemical formulae of the following:
(a) Magnesium chloride (b) Calcium oxide (c) Copper (II) nitrate
(d) Aluminium chloride (e) Calcium carbonate
Q.20 Give the names of the elements present in the following compounds:
(a) Quick lime (b) Hydrogen bromide (c) Baking powder (d) Potassium sulphate
Ans. 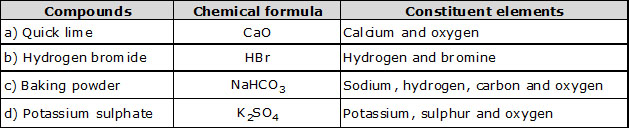
Q.21 Calculate the molar mass of the following substances:
(a) Ethyne, C2H2
(b) Sulphur molecule, S8
(c) Phosphorus molecule, P4 (atomic mass of phosphorus = 31)
(d) Hydrochloric acid, HCl
(e) Nitric acid, HNO3
Ans. (a) Molecular mass of ethyne, C2H2 = 2 × atomic mass of C + 2 × atomic mass of H
= 2 × 12 + 2 × 1 = 24 + 2
= 26 u
Molar mass = 26g/mol
(b) Molecular mass of sulphur molecule, S8 = 8 × atomic mass of S = 8 × 32
= 256 u
Molar mass = 256 g/mol
(c) Phosphorus molecule, P4 = 4 × atomic mass of P
= 4 × 31
= 124 u
Molar mass = 124 g/mol
(d) Molecular mass of hydrochloric acid, HCl = 1 × atomic mass of Cl
= 1 × 1 + 1 × 35.5 = 36.5u
Molar mass = 36.5 g/mol
(e) Molecular mass of nitric acid, HNO3 = 1 × atomic mass of H + 1 × atomic mass of N + 3 × atomic mass of O
= 1 × 1 + 1 × 14 + 3 × 16
= 1 + 14 + 48
= 63 u
Molar mass = 63 g/mol
Q.22 What is the mass of:
(a) 1 mole of nitrogen atoms?
(b) 4 moles of aluminium atoms (Atomic mass of aluminium = 27)?
(c) 10 moles of sodium sulphite (Na2SO3)?
Ans. (a) Mass of 1 mole nitrogen atoms, m = n × M
= 1 × 14 = 14g
(b) Mass of 4 mole aluminium atoms, m = n × M
= 4 × 27 = 108 g
(c) Mass of 10 moles of Na2SO3, m = n × M
M = 2 × atomic mass of Na + 1 × atomic mass of S + 3 × atomic mass of O
= 2 × 23 + 1 × 32 + 3 × 16
= 46 + 32 + 48
= 126
m = 10 × 126
= 1260 g
Q.23 Convert into mole.
(a) 12g of oxygen gas (b) 20g of water (c) 22g of carbon dioxide
Ans. (a) Molar mass of oxygen (M) = 2 × 16 = 32g/mol
Mass of oxygen, m = 12g
Number of moles in 12g of oxygen gas, n = = = mole = 0.375 mole
(b) Molar mass of water, = 1 × 2 + 1 × 16 = 18 g/mol
Mass of H2O,(m) = 20g
(c) Molar mass of CO2(M) = 12 + 2 × 16 = 44 g/mol
Mass of CO2(m) = 22g
Number of moles in 22g CO2(n) = = = 0.5 mole
Q.24 What is the mass of:
(a) 0.2 mole of oxygen atoms? (b) 0.5 mole of water molecules?
Ans. (a) Molar mass of oxygen atoms(M) = 16 g/mol
Mass of 0.2 mole of oxygen atoms, m = n × M = 0.2 × 16 = 3.2 g
(b) 0.5 mole of water molecules = n
Molar mass of water molecule(M) = 2 + 16 = 18 g/mol
\ Mass of 0.5 mole of water(M) = n × M = 0.5 × 18 = 9 gram
Q.25 Calculate the number of molecules of sulphur (S8) present in 16g of solid sulphur.
Ans. Given, 16 grams of sulphur = m
Molecular mass of sulphur (M) = 8 × atomic mass of S
= 8 × 32 = 256 u
\ Molar mass(M) = 256 g/mol
Number of moles of sulphur(n) = = = mole
Number of sulphur molecules = n × Avogadro’s number (N0)
=
= 3.76 × 1022 molecules.
Q.26 Calculate the number of a aluminium ions present in 0.051 g of aluminium oxide.
(Hint: The mass of an ion is the same as that of an atom of the same element. Atomic mass of Al = 27u)
Ans. Mass of aluminium oxide (Al2O3) = m = 0.051g
Molar mass of aluminium oxide, = 2 × 27 + 3 × 16
= 54 + 48 = 102 g/mol
Number of moles of aluminium oxide (n) = =
= 0.5 × 10–3 moles of Al2O3
Number of Al2O3 molecules = 0.5 × 10–3 × 6.022 × 1023
= 3.011 × 1020
Al2O3 ® 2Al3+ + 3O2–
One molecule of Al2O3 gives = 2Al3+ ions
\ 3.011 × 1020 molecules will give = 2 × 3.011 × 1020 Al3+ ions
= 6.022 × 1020 Al3+ ions
Q.1 The elements present in baking soda are -
(A) Sodium, carbon and oxygen (B) Sodium, carbon and hydrogen
(C) Sodium, carbon, hydrogen and oxygen (D) Potassium, carbon and oxygen
Q.2 The first scientist to use of the symbols of elements was-
(A) Dalton (B) Berzillius (C) Kanad (D) Proust
Q.3 The overall charge on an ionic compound is equal to -
(A) Charge of the cation present
(B) Zero
(C) Charge of the anion present
(D) sum of charges of the cation & anion
Q.4 The chemical formula of the copper nitrate -
(A) Cu(NO3)2 (B) CuNO3 (C) Cu2(NO3)3 (D) Cu2NO3
Q.5 The number of carbon atoms in 1g of CaCO3 is-
(A) 6.022 × 1023 (B) 6.022 × 1021 (C) 3.0125 × 1022 (D) 1.204 × 1023
Q.6 The mass of a single atom of carbon is -
(A) 12 g (B) 1/12 g (C) 1.99 × 10–23g (D) 1.99 × 1023g
Q.8 How many molecules are present in 9g of water -
(A) 3.01 × 1023 (B) 6.022 × 1023 (C) 6.08 × 1023 (D) 3.82 × 1023
Q.9 Mg + O2 'X' , 'X' is -
(A) MgO (B) Mg2O (C) MgO2 (D) Mg2O3
Q.10 The formula of sulphuric acid is-
(A) H2SO3 (B) H2SO4 (C) H2SO5 (D) H2S2O7
Q.11 What is true about potassium chlorate -
(A) It gives oxygen gas on strong heating
(B) Its molecular mass is 122.5kg/mol
(C) 122.5g of it contain oxygen atoms three times the Avogadro number
(D) Its molecular formula is KClO4
Q.12 Mass of one Avogadro's number of O atom is equal to -
(A) 16 amu (B) 16g (C) 32g (D) 6kg
Q.13 Hydrogen reacts with oxygen to form water (H2O). The ratio between masses of Hydrogen and oxygen is -
(A) 1 : 8 (B) 63.5 : 8 (C) 2 : 1 (D) 63.5 : 16
Q.14 The correct formula of aluminium sulphate is
(A) AlSO4 ( B) Al2SO4 (C) Al3(SO4)2 (D) Al2(SO4)3
Q.15 Which of the following has highest intermolecular forces of attraction -
(A) Liquid water (B) Liquid ethyl alcohol (C) Gaseous CO2 (D) Solid CO2
Q.16 Which of the following is not correct regarding gases-
(A) Gases exert pressure
(B) Gases have same intermolecular space
(C) Gases have tendency to diffuse
(D) Gases have high intermolecular forces of attraction
Q.17 The Boiling point of water at normal atmospheric pressure is -
(A) 273 K (B) 373 K (C) 100 K (D) 0°C
Q.18 Avogadro's number represents the number, of atoms in-
(A) 12g of C12 (B) 320g of sulphur (C) 32g of oxygen (D) 12.7 g of iodine
Q.19 Molecular mass of ozone is
(A) 16 u (B) 32 u (C) 48 u (D) 64 u
Q.20 Which of the following is not correct according to Dalton's atomic theory?
(A) Matter is made up of atoms
(B) Atoms of all substances are identical in all respects
(C) Atoms combine in a simple whole number ratio
(D) Atoms of two elements can combine to form more than one compound.
Q.21 The temperature at which a liquid changes into gas is called...................
Q.22 Intermolecular space in solid is ................. than that of liquids
Q.23 Change of liquid state to solid state is called ...................
Q.24 Smell of cooked food reaches us in seconds due to the process known as. ...................
Q.25 One mole atoms of oxygen contains .....................atoms of oxygen.
Q.26 The number of atoms in a molecule of the elementary substance called is ...................
Q.27 In water, the proportion of hydrogen and oxygen is .....................by mass.
Q.28 Latin name of mercury is ......................
Q.29 Avogadro number is .........................
Q.30 One mole of sodium sulphate contains ..................... atoms of sodium, ..................... atoms of sulphur and ..................... atoms of oxygen.
Q.31 Intermolecular forces are maximum in ...................
Q.32 Water has boiling point equal to .....................
Q.33 Fusion is a change of ....................to .....................state.
Q.34 match the following elements & compounds given in column-A with column-B
Column-A Column-B
Elements compound Atomicity
(1) Argon (a) 8
(2) Sulphur (b) 4
(3) Oxygen (c) 2
(4) Phosphorous (d) 1
(5) Ozone (e) 3
(6) Bromine (f) 5
(7) Carbon monoxide (g) 6
(8) Hydrogen peroxide (h) 7
(9) Lime water
(10) Ammonia
(11) Quick Lime
(12) Baking Powder
(13) Lime Stone
(14) Common salt
(15) Sodium Sulphate
Objective type questions
1. C 2. A 3. B 4. A
5. B 6. C 7. B 8. A
9. A 10. B 11. C 12. B
13. A 14. D 15. D 16. D
17. B 18. A 19. C 20. B
Fill in the blanks
21. boiling point 22. less
23. solidification 24. diffusion
25. Contain 6.022 × 1023 26. Atomicity
27. 1 : 8 28. Hydragyrum
29. 6.022 × 1023
30. 12.044 × 1023 , 6.022 × 1023, 24.088 × 1023
31. Solid 32. 100°C
33. Solid, liquid
Match the column
1. d 2. a 3. c 4. b
5. e 6. c 7. c 8. b
9. f 10. b 11. c 12. g
13. f 14. c 15. h
Q.1 Name of the building block of all matter.
Q.2 What are the symbols of copper and cobalt ?
Q.3 What is 1 u ?
Q.4 Give symbols for the following elements : Aluminium, Tin, Bromine, Neon.
Q.5 What is ratio between masses of
(i) hydrogen and oxygen in H2O
(ii) nitrogen and hydrogen in NH3?
[Atomic mass of H = 1 u, O = 16 u, N = 14 u]
Q.6 What is meant by formula unit mass?
Q.7 What is meant by valency of an element?
Q.8 10 g silver nitrate solution are added to 10 g of sodium chloride solution. What change in mass do you expect after the reaction?
Q.9 Why is copper represented by the symbol 'Cu' while there is no letter 'u' in the name?
Q.10 What do you understand by the 'atomicity' of the substance?
Q.11 Give two examples each of bivalent cations and bivalent anions.
Q.12 How are mass, molar mass and number of moles related to each other?
Q.13 What is the first law of chemical combination ?
Q.14 What is the conclusion of the Dalton's atomic theory ?
Q.15 Define a molecule. How many molecules are present in
(i) 9g of water (ii) 17g of ammonia
Q.16 What is meant by the term chemical formula ?
Q.17 What are poly atomic ions ? Give example.
Q.18 Calculate the number of molecules of Sulphur (S8) present in 16g of solid sulphur.
Q.19 Give the names of the elements present in the following compounds.
(i) Baking powder (ii) Calcium carbonate (iii) Hydrogen bromide
Q.20 Define the term mole.
Q.21 Write the molecular formula of the following and give the ratio by mass of atoms present ?
(i) Ammonia (ii) Water (iii) Methane
Q.22 A hydrogen is found to contain 14.3% hydrogen and 85.7% carbon. If molecular mass of the hydrocarbon is 28, find out its molecular formula (17g).
Q.23 Write down the names of the compound represented by the following formula :-
(i) Al2(SO4)3 (ii) CaCl2 (iii) K2SO4 (iv) KNO3
Q.24 What is meant by the term chemical formula?
Q.25 Calculate the mass of CO2 which will contain the same number of molecules as are contained in 40.0 g of oxygen.
Q.26 If 1 g of SO2 contains x molecules, what will be the number of molecule in 1 g of methane?
(S = 32 u, O = 16 u, C = 12 u, H = 1 u)
Q.27 What is 'molar volume'? What is its value?
Q.28 Define the "law of constant proportions". How does Dalton's atomic theory explain the truth of the law?
Q.29 What is the difference between the actual mass of a molecule and gram molecular mass?
Q.30 What is formula unit mass? For what type of compounds is it used and why?
Q.31 What is the unit of atomic mass or molecular mass? Define it. What is the mass of this unit in kilograms?
Q.32 State and explain Law of Conservation Mass. How does Dalton's atomic theory explain this law?
Q.33 State and explain the following :
(i) Atom (ii) Molecule (iii) Atomic mass (iv) Molecular mass
Q.34 Arrange the following in order of increasing masses : (i) 0.1 g atom of silver (ii) 0.1 mole of H2SO4 (iii) 1023
molecule of CO2 gas (iv) 1 gram of carbon (v) 1023 atoms of calcium.
(Atomic masses : Ag = 108 u, S = 32 u, N = 14 u, Ca = 40 u)
Q.35 How is the molecular formula of a compound related to its empirical formula?
Q.36 Valencies or the charge numbers of some ions are given below :
Aluminium ion 3+
Magnesium ion 2+
Potassium ion 1+
Nitride ion 3–
Sulphate ion 2–
Fluoride ion 1–
Using the above information, write down the chemical formulae of the following :
(i) Aluminium ion
(ii) Magnesium nitride
(iii) Aluminium sulphate
(vi) Potassium fluoride
(v) Magnesium fluroide
(vi) Potassium nitride
Q.37 The molecular formula of a compound is CH3OH. Calculate its molecular mass in atomic mass units. Also write
down its molar mass. (atomic masses are : H = 1 u, C = 12 u, O = 16 u)
Q.38 Find the number of atoms of each type present in 3.42 grams of canesugar (C12H22O11).
Q.39 What are the postulates and limitations of Dalton's atomic theory?
1. atoms
2. Cu & Co
3. 1 u stands for one twelth (1/12) in the mass of a carbon (carbon-12) atom,
4. Al, Sn, Br, Ne,
5. (i) 1 : 8 (ii) 14 : 3
6. Refer theory
7. Refer theory
8. No change
9. Symbol Cu has been taken from the latin word 'cuprum' which means copper
10. The number of atoms present in one molecule of the substance is called atomicity
11. Cations = Zn2+, Mg2+, anions = SO42–, CO32–
12. Number of moles (n) = or m = n × M.
13. Law of conservation of mass
14. Refer theory
15. (i) 3.011 × 1023 (ii) 6.021 × 1023
16. Refer theory
17. SO4–2, NO3–
18. 3.76 × 1023
19. (i) Na, H, C, O (ii) Ca, C, O (iii) H, Br
20. 1 mole = NA molecule
21. (i) 14 : 3 (ii) 1 : 8 (iii) 3 : 1
22. C2H4
23 (i) Aluminium sulphate
(ii) Calcium chloride
(iii) Potassium sulphate
(iv) Potassium nitrate
25. 110g
26. 4x molecule
34. (i) 10.8 g (ii) 9.8g (iii) 7.306g
(iv) 1g (v) 6.642g
order (i) > (ii) > (iii) > (v) > (iv)
35. n =
36. (i) Al+3 (ii) Mg3N2 (iii) Al2(SO4)3 (iv) KF (v) MgF2 (vi) K3N
37. 32u
Q.1 The law of multiple proportions was proposed by :-
(A) Lavoisier (B) Dalton (C) Proust (D) Ritcher
Q.2 The weight of two elements which combine with each other are in the ratio of their :-
(A) Atomic heights
(B) Atomic volumes
(C) Equivalent weight
(D) Molecular weights
Q.3 180 grams of water conati ns _____ moles :-
(A) 100 (B) 10 (C) 180 (D) 0.01
Q.4 What is the weight of 3 gram atoms of sulphur ?
(A) 96 gm (B) 99 gm (C) 100 gm (D) 3 gm
Q.5 How many gram atoms are present in 144 g of magnesium ?
(A) 12 (B) 6 (C) 100 (D) 144
Q.6 How many moles of oxygen atoms are present in one mole of acetic acid ?
(A) 1 mole (B) 3 moles (C) 2 moles (D) 6 moles
Q.7 What is the number of particles in one mole of a substance ?
(A) 6.023 × 1023 (B) 6.023 × 10–23 (C) 6.023 (D) 3 × 108
Q.8 How many atoms and how many gram atoms are there in 10 grams of calcium ?
(A) 0.25 gram atoms, 6.023 × 10–23 atoms (B) 0.25 gram atoms, 1.50 × 1023 atoms
(C) 0.1 gram atoms, 6.023 × 1023 atoms (D) 0.1 gram atoms, 1.50 × 10–23 atoms
Q.9 Calculate the weight of 0.1 mole of sodium carbonate :-
(A) 1.06 g (B) 11.06 g (C) 10.6 g (D) 1.106 g
Q.10 How many number of moles are present in 540 g of glucose?
(A) 2 moles (B) 3 moles (C) 4 moles (D) 1 moles
Q.11 How much of lime can be obtained by burning 400 g of lime stone ?
(A) 224 g (B) 220 g (C) 400 g (D) 320 g
Q.12 Chemical analysis of a carbon compound gave the following percentage composition by weight of the elements present in it.
Carbon = 10.06%, hydrogen = 0.84%, chlorine = 89.10%. Calculate the empirical formula of the compound :-
(A) C2H2Cl2 (B) CHCl2 (C) CHCl3 (D) C4H4Cl4
Q.13 0.202 g of a carbon compound, on combustion, gave 0.361 g of carbon dioxide and 0.47 g of water. Calculate the percentage composition of carbon :-
(A) 48.76% (B) 8.07% (C) 43.17% (D) 42.17%
Q.14 One mole of CO2 contains :
(A) 1 gram atom of carbon (B) 2 gram atoms of oxygen
(C) 3 gram atoms of carbon and oxygen (D) all of the above
Q.16 The empricial formula of a compound is CH2O. Its molecular weight is 90. Calculate the molecular formula of
the compound. (Atomic weight C = 12, H = 1, O = 16)
(A) C3H7O3 (B) C4H6O3 (C) C3H6O3 (D) C4H2O3
Q.17 Calculate the weight of 2.5 mole of CaCO3 :-
(A) 200 g (B) 230 g (C) 240 g n (D) 250 g
Q.18 How many gram atoms are present in 256 g of O2 ?
(A) 16 (B) 32 (C) 14 (D) 36
Q.19 How many gram atoms are present in 60 g of carbon ?
(A) 6 (B) 10 (C) 16 (D) 5
Q.20 The number of moles present in 20 grams of CaCO3 is :-
(A) 0.1 (B) 0.2 (C) 0.3 (D) 0.25
Q.21 A hydrogen contain 90% of carbon and 10% hydrogen. The empirical formula of the compound is :-
(A) C2H5 (B) C3H2 (C) C3H4 (B) CH3
Q.22 An element has only one type of :-
(A) Molecules (B) Atoms (C) Mixtures (D) Solutes
Q.23 The simplest formula of a compound having 50% of X(atomic weight 10) and 50% of Y(atomic weight 20) is :-
(A) XY (B) X2Y (C) XY3 (D) X2Y2
Q.24 How many moles of glucose (C6H12O6) are present in 5.4 g ?
(A) 0.03 (B) 0.02 (C) 0.01 (D) 0.1
Q.25 Calculate the number of gram atoms present in 8 g of helium :-
(A) 3 (B) 4 (C) 2 (D) 1
Q.26 How many moles are present in 5.3 g of anhydrous sodium carbonate ?
(A) 0.03 (B) 0.04 (C) 0.05 (D) 0.01
Q.27 Calculate the number of moles present in
60 g of NaOH.
(A) 1.2 (B) 1.5 (C) 2.5 (D) 0.15
Q.28 Calculate the weight of nitrogen present in 0.5 moles of NH3.
(A) 8 g (B) 9 g (C) 1 g (D) 7 g
Q.29 Calculate the number of moles present in
7.3 g of HCl.
(A) 0.2 (B) 0.1 (C) 1 (D) 0.02
Q.30 Calculate the weight in gram of 0.9 gram atoms of zinc.
(A) 50.5 g (B) 58.5 g (C) 56.3 g (D) 53.2 g
Q.31 Calculate the weight of 0.4 gram atoms of carbon.
(A) 2.8 g (B) 4.8 g (C) 3.2 g (D) 4.0 g
Q.32 What is the weight of 3 gram atoms of sulphur?
(A) 98 g (B) 99 g (C) 95 g (D) 96 g
Q.33 16 gram of oxygen is equal to :-
(A) 1 gram atom (B) 0.5 gram mole
(C) 2 gram equivalents (D) all of these
Q.34 When a neutral atom is converted into anion its :-
(A) Size increase (B) Size decrease
(C) Atomic number increases (D) Atomic number decreases
Q.35 Which of th e following i s an ionic compound?
(A) HCl (B) Na2O (C) CO2 (D) SO2
Q.36 The bond formed between two atoms of an electronegative element is :-
(A) Electrovalent (B) Covalent (C) Coordinate (D) None of these
Q.37 Ionic compounds :
(A) Have bonds which are directional (B) Conduct electricity in solid state
(C) do not conduct electricity in molten state
(D) Are generally more soluble in polar solvents than in nonpolar solvents
Q.38 Covalent compound commonly exhibit :-
(A) High solubilities in water (B) Low melting points
(C) High electrical conductivity (D) High boiling points
1. B 2. C 3. B 4. A
5. B 6. C 7. A 8. B
9. C 10. B 11. A 12. C
13. A 14. D 15. A 16. C
17. D 18. A 19. D 20. B
21. C 22. B 23. B 24. A
25. C 26. C 27. B 28. D
29. A 30. B 31. B 32. D
33. D 34. A 35. B 36. B
37. D 38. B
1. The law of multiple proportions was proposed by :-
(A) Lavoisier (B) Dalton (C) Preistley (D) Ritcher
2. The weight of two elements which combine with each other are in the ratio of their :-
(A) Atomic heights (B) Atomic volumes (C) Equivalent weight (D) Molecular weights
3. 180 grams of water conatins ________ moles
(A) 100 (B) 10 (C) 180 (D) 0.01
4. What is the weight of 3 gram atoms of sulphur
(A) 96 gm (B) 99 gm (C) 100 gm (D) 3 gm
5. How many gram atoms are present in 144 g of magnesium ?
(A) 12 (B) 6 (C) 100 (D) 144
6. How many moles of oxygen atoms are present in one mole of acetic acid ?
(A) 1 mole (B) 3 moles (C) 2 moles (D) 6 moles
7. What is the number of particles in one mole of a substance ?
(A) 6.023 × 1023 (B) 6.023 × 10–23 (C) 6.023 (D) 3 × 108
8. How many atoms and how many gram atoms are there in 10 grams of calcium ?
(A) 0.25 gram atoms, 6.023 × 10–23 atoms (B) 0.25 gram atoms, 1.50 × 1023 atoms
(C) 0.1 gram atoms, 6.023 × 1023 atoms (D) 0.1 gram atoms, 1.50 × 10–23 atoms
9. Calculate the weight of 0.1 mole of sodium carbonate :-
(A) 1.06 g (B) 11.06 g (C) 10.6 g (D) 1.106 g
10. How many number of moles are present in 540 g of glucose?
(A) 2 moles (B) 3 moles (C) 4 moles (D) 1 moles
11. How much of lime can be obtained by burining 400 g of lime stone ?
(A) 224 g (B) 220 g (C) 400 g (D) 320 g
12. Chemical analysis of a carbon compound gave the following percentage composition by weight of the elements present in it.
Carbon = 10.06%, hydrogen = 0.84%, chlorine = 89.10%. Calculate the empirical formula of the compound :-
(A) C2H2Cl2 (B) CHCl2 (C) CHCl3 (D) C4H4Cl4
13. 0.202 g of a carbon compound, on combustion, gave 0.361 g of carbon dioxide and 0.47 g of water. Calculate the percentage composition of carbon :-
(A) 48.76% (B) 8.07% (C) 43.17% (D) 42.17%
14. One mole of CO2 contains :
(A) 1 gram atom of carbon (B) 2 gram atoms of oxygen
(C) 3 gram atoms of carbon and oxygen (D) all of the above
15. The percentage of nitrogen in ammonia is given by the expression :-
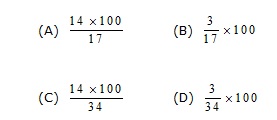
16. The empricial formula of a compound is CH2O. Its molecular weight is 90. Calculate the molecular formula of the compound. (Atomic weight C = 12, H = 1, O = 16)
(A) C3H7O3 (B) C4H6O3 (C) C3H6O3 (D) C4H2O3
17. Calculate the weight of 2.5 mole of CaCO3
(A) 200 g (B) 230 g (C) 240 g (D) 250 g
18. How many gram atoms are present in 256 g of O2 ?
(A) 16 (B) 32 (C) 14 (D) 36
19. How many gram atoms are present in 60 g of carbon ?
(A) 6 (B) 10 (C) 16 (D) 5
20. The number of moles present in 20 grams of CaCO3 is :-
(A) 0.1 (B) 0.2 (C) 0.3 (D) 0.25
21. A hydrogen contain 90% of carbon and 10% hydrogen. The empirical formula of the compound is :-
(A) C2H5 (B) C3H2 (C) C3H4 (B) CH3
22. An element has only one type of :-
(A) Molecules (B) Atoms (C) Mixtures (D) Solutes
23. The simplest formula of a compound having 50% of X(atomic weight 10) and 50% of Y(atomic weight 20) is :-
(A) XY (B) X2Y (C) XY3 (D) X2Y2
24. How many moles of glucose (C6H12O6) are present in 5.4 g ?
(A) 0.03 (B) 0.02 (C) 0.01 (D) 0.1
25. Calculate the number of gram atoms present in 8 g of helium :-
(A) 3 (B) 4 (C) 2 (D) 1
26. How many moles are present in 5.3 g of anhydrous sodium carbonate ?
(A) 0.03 (B) 0.04 (C) 0.05 (D) 0.01
27. Calculate the number of moles present in 60 g of NaOH.
(A) 1.2 (B) 1.5 (C) 2.5 (D) 0.15
28. Calculate the weight of nitrogen present in 0.5 moles of NH3.
(A) 8 g (B) 9 g (C) 1 g (D) 7 g
29. Calculate the numbe of moles present in 7.3 g of HCl.
(A) 0.2 (B) 0.1 (C) 1 (D) 0.02
30. Calculate the weight in gram of 0.9 gram atoms of zinc.
(A) 50.5 g (B) 58.5 g (C) 56.3 g (D) 53.2 g
31. Calculate the weight of 0.4 gram atoms of carbon.
(A) 2.8 g (B) 4.8 g (C) 3.2 g (D) 4.0 g
32. What is the weight of 3 gram atoms of sulphur ?
(A) 98 g (B) 99 g (C) 95 g (D) 96 g
33. 16 gram of oxygen is equal to :-
(A) 1 gram atom (B) 0.5 gram mole
(C) 2 gram equivalents (D) all of these
34. When a neutral atom is converted into anion its :-
(A) Size increase (B) Size decrease
(C) Atomic number increases (D) Atomic number decreases
35. Which of the following is an ionic compound
(A) HCl (B) Na2O (C) CO2 (D) SO2
36. The bond formed between two atoms of an electronegative element is :-
(A) Electrovalent (B) Covalent (C) Coordinate (D) None of these
37. Ionic compounds :
(A) Have bonds which are directional (B) Conduct electricity in solid state
(C) do not conduct electricity in molten state
(D) Are generally more soluble in polar solvents than in nonpolar solvents
38. Covalent compound commonly exhibit :-
(A) High solubilities in water (B) Low melting points
(C) High electrical conductivity (D) High boiling points
1. B 2. C 3. B 4. A
5. B 6. C 7. A 8. B
9. C 10. B 11. A 12. C
13. A 14. D 15. A 16. C
17. A 18. D 19. A 20. D
21. B 22. C 23. B 24. B
25. A 26. C 27. C 28. B
29. D 30. A 31. B 32. B
33. D 34. D 35. A 36. B
37. D 38. A