The compounds obtained from 'Carbon' are widely used as clothes, medicines, books, food, fertilizer, fuel etc. all living structures are carbon based.
The amount of carbon present in the earth's crust and in the atmosphere is quite merge. The earths crust has only 0.02% carbon in the form of
mineral (like carbonates, hydrogen-carbonates, coal and petroleum) and the atmosphere has 0.03% of carbon dioxide. In spite of this small
amount of carbon available in nature, the importance of carbon seems to be immense.
Carbon forms a large number of compounds with hydrogen which are known as hydrocarbons. In addition to hydrogen,
carbon compound may also contain some other element such as oxygen, halogen, nitrogen, phosphorus, sulphur etc.
The number of compounds of carbon is more than three million which is much larger than the compounds formed by all other element put together.
Carbon forms covalent bonds in its compounds with other atoms. In each compound the valency of carbon is four. That is, carbon has tetravalent character.
But what is covalent bond and what is meaning of tetravalent ?
A chemical bond formed between two atoms of the same element or two atoms of different elements by sharing of electron is called a covalent bond.
Necessary conditions of the formation of covalent bond :
* The combining atoms should have nonmetallic character.
* The combining atoms should contain 4 to 7 electrons in their respective valence shell.
* In hydrogen there is only 1 valence electron, but it also forms covalent bond.
* The combining atoms need 1, 2, 3 or 4 electrons to complete their octet (hydrogen completes its duplet)
* The combining atoms should contribute equal number of electrons to form pair of electrons to be shared.
* After sharing the pair of electrons each combining atoms should attain stable electronic configuration like its nearest noble gas.
On the basis of the number of electrons shared by two combining atoms, the covalent bond are of three types.
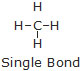
* Single Covalent Bond : A single covalent bond is formed by the sharing of one pair of electrons between the two atoms.
It is represented by one short line (–––) between the two atoms.
Example : H–H, Cl –Cl, H–Cl, CH3–CH3.
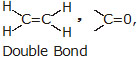
* Double Covalent Bond : A double covalent bond is formed by the sharing of two pairs of electron between the two combining atoms.
It is represented by putting (=) two short lines between the two bonded atoms.
Examples : O = O (O2), CO2 (O = C = O), H2C = CH2
* Triple covalent bond : A triple bond is formed by the sharing of three pair of electrons between the two combining atoms.
It is represented by putting three short line (º) between two bonded atoms.
Example : N2 (NºN), CHºCH.
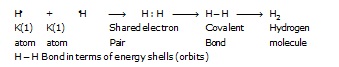
* Formation of hydrogen molecule (H2) :
A molecule of hydrogen is composed of two H-atoms. The electronic configuration of H-atom is.
Shell - K
Electron-1 incomplete duplet (unstable)
Electronic configuration of He atom
Shell - K
Electrons-2 complete duplet (stable)
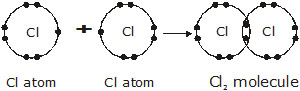
* Formation of chlorine molecule (Cl2). The atomic number of chlorine is 17,
thus there are 17 electrons in an atom of chlorine.
Electronic configuration of Cl atom –
Electronic configuration of Ar atom –
Chlorine atom needs one electron more to complete its octet –
![]()
Cl – Cl bond in terms of energy shell orbits
* H atom has one valence electron. It needs 1 electron more to complete its duplet and chlorine atom has 7 valence electrons.
It need 1 electron more to complete its octet and acquire stable electronic configuration (2, 8, 8) like noble gas argon.
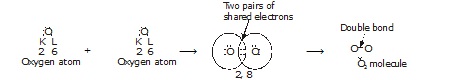
* Formation of oxygen (O2) :
The atomic number of O atom is 8. There are 6 electron in the valence shell of oxygen atom it needs 2 more electrons
to attain the nearest stable inert gas Neon (2, 8) configuration :
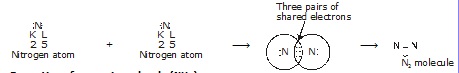
* Formation of nitrogen molecule (N2) :
The atomic number of nitrogen is 7 and its electronic configuration is K(2), L(5). It needs 3 electrons more to
complete its octet like noble gas neon (2, 8).
* Formation of ammonia molecule (NH3) :
The atomic number of N is 7. It's electronic configuration is 2, 5 there are 5 electrons in its valence shell.
It needs 3 electrons more to complete its octet like noble gas neon (2, 8).
* Formation of H2O molecule :
The electronic configuration of hydrogen is K (1) and that of oxygen is K(2) L(6) thus each hydrogen require
one and oxygen required two electrons to achieve the stable electronic configuration.
* Formation of CO2 molecule :
The atomic number of C is 6 and the electronic configuration of C is K(2), L(4) and that of oxygen is K(2),
L(6) thus each carbon require 4 and oxygen require two electrons to achieve the stable electronic configuration.
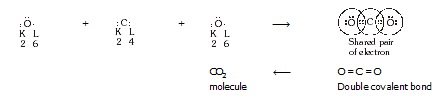
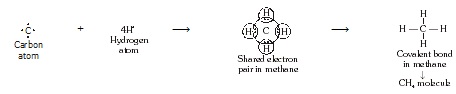
* Formation of CH4 molecule :
Methane is a covalent compound containing 4 covalent bond. It contains one carbon atom and four hydrogen
atom covalently bonded to central carbon atom.
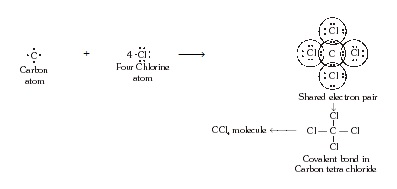
* Formation of carbon tetrachloride molecule (CCl4) :
The electronic configuration of carbon and chlorine atoms are (2, 4) and (2, 8, 7) respectively.
Carbon atom needs four electrons and chlorine atom needs one electron to attain the stable electronic configuration.
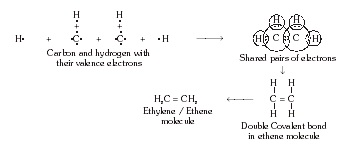
* Formation of ethylene or ethene molecule (C2H4) :
The electronic configuration of carbon atom is 2, 4. There are 4 valence electrons in one C atom.
Each H atom contains 1 valence electron. Thus, there are 12 valence electrons present in ethene molecule.
* Formation of Acetylene or ethyne molecule (C2H2) :
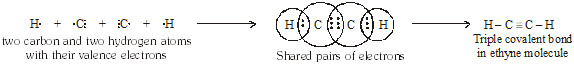
Q. What would be the electron dot structure of carbon dioxide which has formula CO2 ? [NCERT]
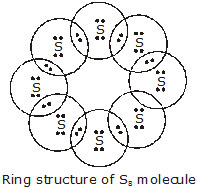
Q. What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur?
Ans. The eight atoms of sulphur are joined together in the form of a puckered ring. [NCERT]
Q. Explain the nature of the covalent bond using the bond formation in CH3Cl. [NCERT]
Q. Draw the electron dot structure for [NCERT]
(a) Ethanoic acid (b) H2S (c) Propanone (d) F2
Non polar and polar covalent compounds:
Non polar covalent bond :
A covalent bond formed between two atoms of the same element or same electronegativity is called a non-polar covalent bond.
xample : H2, N2, O2, Cl2 etc.
Polar covalent bond :
The covalent bond between the atoms of two elements having different electronegativities is called a polar covalent bond. Molecule in which the atom are bonded by a polar covalent bond are called polar molecules.
Note : In a polar covalent bond, the shared pair of electrons lies more toward the atom which is more electronegative.
Example : HCl, H2O & NH3
Note : d means partial
Note: There are some exception in which central atom has less than eight electrons
* Covalent bond are formed by mutual sharing of electrons
Note : Shared pair of electrons is also called bonding pair of electrons.
* Covalent bond is directional in nature because shared pair of electrons remain localized in a definite space between the two atoms.
Characteristics of covalent compounds :
Physical State : The covalent compounds are generally gases or liquids, but compounds with high molecular masses are solids.
Example : Solid : Urea, Glucose, Naphthalene.
Liquids : Water, ethanol, benzene.
Gases : Methane, chlorine, hydrogen, oxygen
* Melting and boiling points : Covalent compounds have low melting and low boiling points because intermolecular forces
(cohesive forces) in covalent compounds are weaker than those in ionic compounds.
Note : Some exception like diamond and graphite which are covalent solids have very high M.P. & B.P.
* Solubility :
Covalent compounds generally dissolve readily in organic solvents but they are less soluble in water.
For example : Napthalene which is an organic compound dissolves readily in organic solvents like ether but is insoluble in water.
However some covalent compounds like urea, glucose, sugar etc. are soluble in water. Some polar covalent compounds like
ammonia and hydrochloric acid are soluble in water.
* Conductivity :
Covalent compounds do not conduct electricity because they contain neither the ions nor free electrons necessary for conduction, So they do not conduct electricity
For example : Covalent compounds like glucose, alcohol, carbon tetrachloride do not conduct electricity.
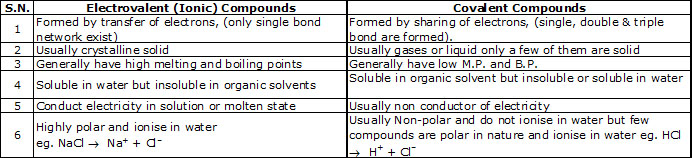
Differences between ionic and covalent compounds :
The chemical compounds which are present in living organisms (plant and animal) are called organic compounds.
The belief that formation of organic compounds was possible only in plants and animals led the scientists of early
days to propose that Vital Force was necessary for the formation of such compounds. But the experimental work
of Friedrich Wohler (German chemist) denied the idea of vital force when he preparedurea in his laboratory.
(urea is an organic compound and waste product of urine).
Q. Name the organic compound which was prepared by Wohler in his laboratory. [NCERT]
Allotropy / allotropes of carbon :
The phenomenon of existence of allotropic forms of an element is called allotropy. Allotrops are the different forms
of the same element having different physical properties but almost similar chemical properties.
There are three allotrops of carbon these are diamond, graphite and fullerene.
Diamond : Diamond is a crystalline allotrope of carbon. Its atomic symbol & empirical formula is 'C'.
Structure of Diamond
Structure : In diamond, each carbon atom is covalently bonded to four other carbon atoms in a tetrahedral arrangement.
This tetrahedral arrangement of carbon atoms gives a rigid, three dimensional structure to diamond
It is due to this rigid structure that diamond.
* Is very hard crystalline structure.
* Has high melting point.
* Is non conductor of heat and electricity.
Properties : Pure diamond is a transparent and colourless solid.
* Polished diamond sparkles brightly because it reflects most of the light (refractive index of diamond is 2.45)
* Diamond are not attacked by acids, alkalis and solvents like water, ether, benzene or carbon tetrachloride but
diamond is attacked by fluorine at 750°C.
* The density of diamond is 3.51 g per cm3 at 20°C.
Uses :
* A saw fitted with diamond is used for sawing marbles.
* A chip diamond is used for glass cutting.
* Black diamonds are used in making drill.
* Diamonds are used for making dice for drawing very thin wires of harder metals.
* Diamonds are also used for making high precision tools for use in surgery such as, for the removal of cataract.
* Diamond are used for making precision thermometers and protective windows for space crafts.
Graphite :
Graphite is also known as black lead it marks paper black. The name graphite has been taken from the Greek
word ''graphein" (which means to write) in reference to its uses as 'lead' in lead pencils.
Structure :
Graphite is an opaque and dark grey solid. In a crystal of graphite the carbon atoms are arranged in hexagonal
patterns in parallel planes. In a layer of graphite each carbon atom is strongly bonded to three carbon atoms by covalent bonds.
Thus, one valence electron of each carbon atom is free in every layer of graphite crystal. The free electron makes graphite a good conductor of electricity.
Each layer is bonded to the adjacent layers by weak forces. As a result, each layer can easily slide over the other.
Properties :
* Graphite is greyish -black, opaque material having metallic (shiny) lustre.
* It is soft and has a soapy (slippery) touch.
* Graphite is lighter than diamond. The density of graphite is 2.26 g per cm3 at 20°C.
* Graphite is a good conductor of heat and electricity.
* Graphite has a very high melting point.
* Graphite is insoluble in all common solvent.
Uses :
* For making electrodes in dry cells and electric arc furnaces.
* Graphite is a good dry lubricant for those parts of machines where grease and oil cannot be used.
* For making crucibles for melting metals.
* For manufacturing lead pencils.
* Graphite is used as neutron moderator in nuclear reactors.
* For the manufacture of gramophone records and in electrotyping.
* For the manufacture of artificial diamond.
Fullerene :
* Fullerene was discovered in 1985 by Robert F. Curl Jr, Harold Kroto and Richard E. Smally.
* This molecule containing sixty atoms of carbon has been named Buckminster fullerene.
Fullerens has been named after American architect and engineer R. Buckminster-fuller
whose geodesic domes follow similar building principles.
Types of fullerene :
C60, C70, C74 and C78 are the members of the fullerene family. But C60 is the most stable and most studied form of fullerenes.
Structure of fullerene :
* Buckminster fullerene molecule (C60) is nearly spherical.
* It consists of 12 pentagonal faces and 20 hexagonal faces giving it 60 corners. Thus, Buckminster fullerene has a hollow, cage-like structure.
* In figure, ball like molecules containing C atoms.
Preparation :
* By electrically heating a graphite rod in atmosphere of helium.
* By vaporising graphite by using laser.
Properties :
* Fullerene is soluble in benzene and forms deep violet colour solution.
* Crystalline fullerene has semiconductor properties.
* Compounds of fullerene with alkali metals are called fullerides and they are superconductors.
Uses :
* As a superconductor.
* As a semiconductor.
* As a lubricants and catalyst.
* As fibres to reinforce plastics.
About three million (or thirty lakh) compounds of carbon are known. The existence of such a large number of organic
compounds is due to the following characteristic features of carbon.
(1) CATENATION : Tendency to form Carbon-Carbon bond :
"The property of forming bonds with atoms of the same element is called catenation".
Carbon has the maximum tendency for catenation in the periodic table. This is because of strong carbon carbon bonds as compared to other atoms.
Note: Silicon also show catenation.
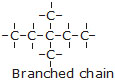
* When two or more carbon atoms combine with one another, they form different types of chain such as
(i) Straight chains
(ii) Branched chains
(iii) Closed chain or ring chains
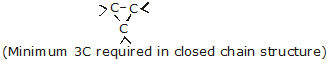
* The property of catenation is due to
(i) small size of C atom (ii) Great strength of carbon - carbon bond.
(2) Tetravalency of Carbon :
* The atomic number of carbon is 6.
* The electronic configuration of carbon atom is 1s2,2s2,2p2.
* It has four electrons in the outermost shell, therefore its valency is four. Thus carbon forms four covalent bonds in its compounds.
(3) Tendency to form multiple bonds :
Due to small size, carbon can easily form double or triple bonds (called multiple bonds) with itself and with the atoms of other elements as nitrogen, oxygen, sulphur etc.
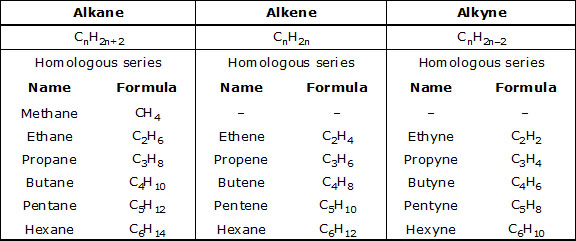
"A series of organic compounds having similar structures and similar chemical properties in which the successive members differ in their molecular formula by –CH2 group".
The different members of the series are called homologous.
Characteristics of Homologous Series :
* All the member of a homologous series can be described by a common general formula.
Example : All alkane can be described by the general formula CnH2n+2.
* Each member of a homologous series differ from its higher and lower neighbouring members by a common difference of –CH2 group.
* Molecular masses of the two adjacent homologues differ by 14 mass units, because molecular mass of –CH2 group is 12 + 2 = 14.
* All the members of a homologous series show similar chemical properties.
* All the members of the series can be prepared by similar methods known as the general method of preparation.
Table : Some members of alkane, alkene and alkyne homologous series.
Activity : Calculate the difference in the formulae and molecular masses for (a) CH3OH and C2H5OH (b) C2H5OH and C3H7OH and (c) C3H7OH and C4H9OH
Q. It there any similarity in these three ? [NCERT]
Q. Arrange these alcohols in the order of increasing carbon atoms to get a family. Can we call this family a homologous series ? [NCERT]
Q. What is homologous series? Explain with an example. [NCERT]
Solution :
Conclusion :
(i) Yes, all these compounds are the members of a homologous series for alcohols.
(ii) CH3OH, C2H5OH, C3H7OH and C4H9OH -increasing carbon atoms. These four compounds form a
homologous series.
* Aldehydes :
HCHO, CH3CHO, CH3CH2CHO, CH3CH2CH2CHO
* Carboxylic acids : HCOOH, CH3COOH, CH3CH2COOH,CH3CH2CH2COOH
* Amines : CH3NH2, CH3CH2NH2,CH3CH2CH2NH2.
* Ketones : CH3COCH3, CH3COCH2CH3, CH3COCH2CH2CH3
* Haloalkanes : CH3X, CH3CH2X, CH3CH2CH2X, CH3CH2CH2–CH2X
How do physical properties change in a homologous series of hydrocarbons.
The physical properties of the various members of a homologous series change regularly with an increases in the molecular mass.
(i) Melting and boiling points : Melting point and boiling point of hydrocarbon in a homologous series increases with an increase in molecular mass.
(ii) Physical State :
* Hydrocarbons containing lesser number of carbon atoms are gases.
* Hydrocarbons containing large number of carbon atoms are solids.
* Hydrocarbon containing intermediate number of carbon atoms are liquid.
Example : Hydrocarbon containing 1-4 carbon atoms are gases, these containing 5–13 carbon atoms are liquid and those containing more than 14 carbon atoms are solids.
Compounds formed from combination of carbon and hydrogen are known as hydrocarbon. Hydrocarbon on the basis of chain are mainly classified into two parts.
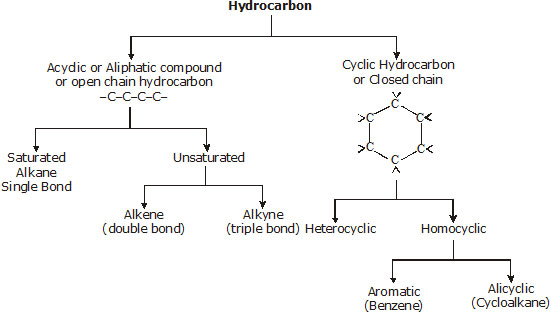
(1) Saturated Hydrocarbon :
* The hydrocarbons which contain only single carbon-carbon covalent bonds are called saturated hydrocarbons.
* They are also called alkanes.
* General formula for alkanes is CnH2n+2 where 'n' is the number of carbon atoms.
General formula of saturated hydrocarbon (CnH2n+2)
(2) Unsaturated hydrocarbons :
The hydrocarbon in which two carbon atoms are bonded to each other by a double (=) or a triple (º) bond is called an unsaturated hydrocarbon.
* Unsaturated hydrocarbons are of two types viz. alkenes and alkynes.
(I) Alkenes : ()
* The hydrocarbon in which the two carbon atoms are bonded by a double bond are called alkenes.
* Their general formula is CnH2n where "n" is the number of carbon atoms.
General formula of alkenes : CnH2n
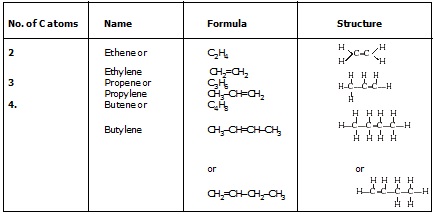
(II) Alkyne (– C º C –)
l The hydrocarbon in which two carbon atoms are bonded by a triple bond are called alkyne.
l Their general formula is CnH2n–2 where 'n' is the number of carbon atoms.
General formula of alkynes : CnH2n–2
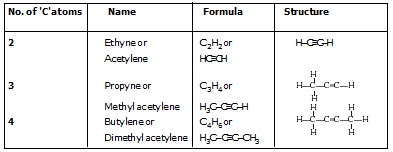
Q. Give a test that can be used to differentiate chemically between butter and cooking oil.[NCERT]
The hydrocarbons may also have branched, closed chains or ring or cyclic structures.
Branched structure :
The alkanes containing three or less carbon atoms do not form branches.
CH4 CH3–CH3 CH3–CH2–CH3
Methane Ethane Propane
* The alkane containing four carbon atoms (C4H10) has two types of arrangement of carbon atoms.
* Closed chains or cyclic hydrocarbon :
These hydrocarbons contains closed chain or rings of atoms in their molecules. These are of two types:
(A) Alicyclic hydrocarbon :
* These hydrocarbons contain a ring chain of three or more carbon atoms.
* These cyclic compounds are named by prefixing cyclo before the name of corresponding straight chain hydrocarbon.
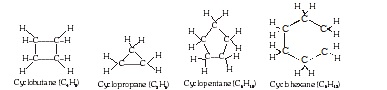
(B) Aromatic hydrocarbon :
* These have at least one benzene ring in their molecules.
* It is a special type of ring of six carbon atoms with three double bonds in alternate positions.
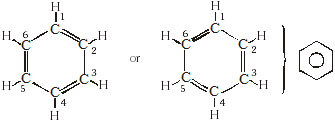
* Carbon forms many compounds with hydrogen. But carbon also forms bonds with other atoms such as halogen,
oxygen, nitrogen and sulphur. Therefore, carbon is said to be very friendly element.
These compounds are obtained by replacing one or more hydrogen atoms by other atoms such that the valency
of carbon remains satisfied. The atom replacing the hydrogen atom is called heteroatom or Functional group.
* Different organic compounds having same functional group have almost same properties these are called families.
Example :
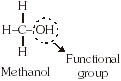
* Properties of CH3–OH and CH3–CH2OH are similar and it is due to the presence of –OH (hydroxyl) group.
* This group is known as alcoholic group.
* Family of compounds having –OH group is called alcohols.
Some Functional Groups in Carbon compounds
Nomenclature of carbon compounds :
Carbon compounds can be called by their common names, but, then remembering millions of compounds by their
individual names may be very difficult. Due to this reason, the International Union of Pure and Applied chemistry
(IUPAC) has devised a very systematic method of naming these compounds.
Naming a carbon compound can be done by the following methods.
* The number of carbon atoms in the molecule of a hydrocarbon is indicated by the following stems.
Example : Saturated hydrocarbon.
Alkane ® Meth + ane = Methane
Unsaturated hydrocarbon
Alkene ® Eth + ene = Ethene
Alkyne ® Eth + yne = Ethyne
* In case of functional group is present, it is indicated in the name of compound with either a prefix or a suffix.
* Identify the longest continuous chain of carbon atoms. This gives the name of parent hydrocarbon.
* In the case of any substituent appropriate prefix is added before the name of parent hydrocarbon.
* In the case of a functional group, the ending 'e' in the name of the parent hydrocarbon is replaced by the appropriate suffix.
"Functional group may be define as an atom or a group of atoms which is responsible for most of the characteristic chemical properties of an organic compound".
The prefixes and suffixes of some substituents/functional group
Q. How many structure isomers can you draw for pentane ?
Q. What will be the formula and electron dot structure of cyclopentane ?
Q. Draw the structure for the following compounds :
(i) Ethanoic acid
(ii) Bromopentane
(iii) Butanone
(iv) Hexanal
Q. Draw the possible structural isomers for bromopentane.
Q. How would you name the following compounds ?
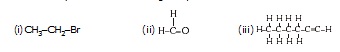
Definition: Compounds having same molecular formula show different physical and chemical properties are
called isomers and the phenomenon is called isomerism.
The difference in properties of isomers is due to the difference in the relative arrangements of various atoms
present in their molecules. Organic compound show following types of structural isomerism on the basis of
their difference in structural arrangement of atoms.
® Type of Isomerism: The following figure shows the pictorial representation of different types of isomerism
® Chain isomerism: Organic compounds having same molecular formula but difference in the
nature of length carbon chain are called chain isomers.
For example, Let us consider the molecular formula of an alkane C4H10.
® Position isomerism: Compounds having same molecular formula but differ in the position of, functional group,
double bond or triple bond in the carbon chain are called position isomers.
This type of isomerism is shown by Alkene, Alkyne, Alcohol, Amine, Haloalkane etc.
Ex.1 Let us consider the molecular formula of an alcohol (CnH2n + 2O)C3H8O
Ex.2 Let us consider the molecular formula of alkene C4H8(CnH2n)
® Functional isomerism: Compounds having same molecular formula but differ in nature of functional group are called functional isomers.
Ex.1 Alcohol and ether have same molecular formula (CnH2n+2O) but have different functional group hence show functional isomerism. C2H6O
Ex.2 Aldehyde and Ketone having same molecular formula (CnH2nO)C3H6O
® Metamerism: The compounds having same molecular formula but different number of carbon atoms
(or alkyl groups) on either side of functional group, are called metamers.
E.g. ethers, thioethers, secondary amines, ketones, esters etc.
(a)
(b) 
® Tautomerism: This is a special type of functional isomerism in which the isomers differ in the arrangement of atoms but they
exist in dynamic equilibrium with each other. For exmaple, acetaldehyde and vinyl alcohol are tautomers.
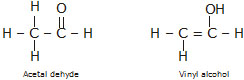
Stereoisomerism was exhibit by compounds which have same structural formula and sequence of bonds but differ
in the relative position of atoms or groups of atoms in space. It is majorly of two types
1. Geometrical Isomerism 2. Optical Isomerism
1. Geometrical Isomerisms also called cis trans isomerism is exhibited by alkanes because of the presence of double bond.
This is due to the restricted rotation around carbon-carbon double bond. As a result, the position of the groups attached to
these carbons is fixed in space. Here cis-isomer have identical group on either side where as intrans-isomer identical
groups are at opposite side of C = C.
2. Optical Isomerism is shown by substances which can rotate the plane of polarished light.
For exmaple this type of isomerism is shown by amino acid alanine.
Note: You will study about stereoisomerism in detail in higher classes.
All carbon compounds show some common characteristic properties. As most of the fuels we use are either carbon
or its compounds. Some such properties are described here :
Combustion :
Combustion is a chemical process in which heat and light (in the form of flame) are given out
The process of combustion, is a rapid oxidation reaction of any substance in which heat and light are produced.
l Combustion of Carbon : Carbon (or charcoal) burn in air or oxygen to give CO2 producing heat and light.
![]()
Q. Why carbon and its compounds are used as fuels for most applications?
l Combustion of Hydro Carbon : Hydrocarbons burn to produce carbon dioxide (CO2), water (H2O) and heat and light.
![]()
Note : Natural gas and biogas contain methane. So, burning of natural gas and biogas are also combustion reactions.
Burning of LPG (Butane) produces CO2, H2O heat and light.
![]()
l Combustion of cellulose :
Combustion of cellulose (like wood, cotton cloth and paper) gives CO2, H2O heat and light. Cellulose is a carbohydrate
and can be described by the formula (C6H10O5)n.
![]()
l Combustion of alcohol :
![]()
Activity : To observe the combustion of given organic compounds.
Materials : Benzene, naphthalene, Camphor, alcohol (ethanol). Spirit, acetone.
Procedure :
1. Take each compound on iron spatula and burn them in Bunsen burner.
2. Record the type of flame produced.
3. Put a metal plate above the flame and observe whether or not there is black carbon deposition.
Compound used Flame Produced Deposit
Benzene Smoky flame Carbon deposited
Naphthalene Smoky flame Carbon deposited
Camphor Smoky flame Carbon deposited
Alcohol Non-Luminous flame No Carbon deposited
Spirit Non-Luminous flame No Carbon deposited
Acetone Non-Luminous flame No Carbon deposited
Benzene, naphthalene, camphor burn with smoky flame and carbon particles get deposited they undergo
incomplete combustion due to excess of carbon content.
l Alcohol, spirit and acetone burn with non-Luminous flame and no carbon gets deposited.
They under go complete combustion, therefore produce more heat.
Activity : To study the different types of flames / presence of smoke.
Material required : Bunsen burner.
Procedure :
1. Light the bunsen burner.
2. Close the air hole and observe the colour of the flame.
3. Put a metal plate over it and observe the nature of deposit.
4. Open the air regulator to allow flow of air.
5. Observe the colour of flame.
6. Put a metal plate and observe the nature of deposit.
Observation :
Conclusion : Keep the air regulator open to get oxidising, non-sooty flame which has high temperature and does not lead to black deposits.
Combustion and the nature of flame :
(i) Saturated hydrocarbon such as, methane, ethane, propane, butane and natural gas and LPG burn
with a blue flame in the presence of sufficient / excess of air / oxygen.
(ii) In the presence of limited amount of air / oxygen, saturated hydrocarbon, such as, methane, butane. etc give smoky flame.
(iii) Unsaturated hydrocarbon such as ethene, ethyne etc. burn with a luminous / yellow smoky flame.
(iv) The gas / kerosene stove used at home has inlets for air so that a sufficiently oxygen rich mixture is burnt to give a
clean blue flame. If you carefully observe the bottoms of vessels getting blackened, it is clear indication that the air holes
are blocked and the fuel is getting wasted.
(v) Fuels, such as coal and petroleum, have some amount of nitrogen and sulphur in them.
Combustion of coal and petroleum results in formation of oxides of sulphur and nitrogen
(such as sulphur dioxide, nitric oxide, nitrogen peroxide) which are major pollutants in the environment.
Coal and petroleum have been formed from biomass which has been subjected to various biological and geological processes.
Coal is a naturally occurring black mineral and is a mixture of free carbon and compounds of carbon containing hydrogen,
oxygen, nitrogen and sulphur. It is not only a good fuel but is also a source of many organic compounds.
It is found in coal mines deep under the surface of earth.
Coal is believed to be formed from fossils which got buried inside the earth during earthquakes and volcanoes which
occurred about 300 million years ago. Due to huge pressure and temperature inside the earth and in the absence of air,
the fossils fuels (vegetable matter or wood, etc.) were converted into coal. The slow chemical processes of the conversion
of wood into coal is called carbonization. Since coal is formed by slow carbonization of plants and fossils,
it produces many important carbonisation products like peat, lignite, bituminous and anthracite etc. and is itself known as fossil fuel.
Coal is also a non-renewable source of energy.
Petroleum is a complex mixture containing various hydrocarbons (compounds of carbon and hydrogen) in addition to small amounts
of other organic compounds containing oxygen, nitrogen, and sulphur. It is a dark coloured, viscous and foul smelling crude oil.
The name petroleum is derived from latin words : "petra" meaning rock and "oleum" meaning oil.
Since petroleum is found trapped between various rocks, it is also known as rock oil.
Carbon and its compounds can be easily oxidised on combustion (or burning). During combustion / burning, the compounds
gets oxidised completely to different products, depending upon the nature of the oxidising agents.
l Carbon gives carbon monoxide or carbondioxide depending upon the oxygen available.
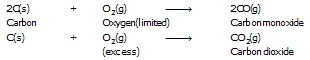
l Hydrocarbon when oxidised give different product as follows :
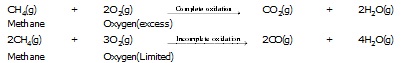
l Alcohols also give different products on oxidation depending upon the reaction conditions.
Example :
Alcohols on oxidation with certain oxidising agents such as chromic anhydride in acetic acid, yield corresponding aldehydes,
where as on oxidation with alkaline potassium permanganate (or acidified potassium dichromate)
corresponding carboxylic acid is formed, as given below :
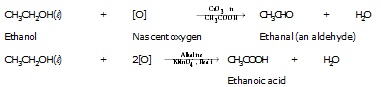
Activity : To study the reaction of ethanol with alkaline potassium permanganate :
Material required : Ethanol, alkaline KMnO4, test tube.
l Take about 3 ml of ethanol in a test tube.
l Add 5% solution of alkaline KMnO4 drop by drop into this solution.
l Observe the colour of alkaline KMnO4 after adding initially as well as finally.
Observation : The colour of KMnO4 gets discharged in the beginning. When excess of KMnO4 is added,
the colour of KMnO4 does not disappear because whole of ethanol gets oxidised to ethanoic acid.
![]()
All unsaturated hydrocarbons (unsaturated carbon compounds) react with a molecule like H2. X2. H2O etc. to form another saturated compounds are called addition reactions.
Unsaturated hydrocarbons add hydrogen, in the presence of catalysts, such as nickel or palladium to give saturated hydrocarbons.
Note : Catalysts are substance that cause a reaction to occur or proceed at a different rate without the reaction it say being affected.
l Addition of hydrogen to ethene :
Q. Which of the following hydrocarbons undergo addition reactions : C2H6, C3H8, C3H6, C2H2 and CH4.
[NCERT]
l Addition of hydrogen to a unsaturated carbon compound is called hydrogenation reaction.
Certain vegetable oils such as ground nut oil, cotton seed oil and mustard oil, contain double bonds
(C = C) and are liquids at room temperature. Because of the unsaturation, the vegetable oils undergo hydrogenation,
like alkenes, to from saturated products called vanaspati ghee. Which is semi-solid at room temperature.
Vegetable oils (Unsaturated oil) + Hydrogen Vanaspati Ghee (Saturated ghee)
Q. What is hydrogenation? What is its industrial application? [NCERT]
Q. If a molecule Y contain two – C = C – double bonds, then how many moles of H2 are required for complete hydrogenation of one mole of Y? [NCERT]
Q. Write the industrial application of hydrogenation. [NCERT]
Substitution reactions :
The reactions in which one or more hydrogen atoms of a hydrocarbon are replaced by some other atoms or groups are called substitution reaction.
Example :
Methane reacts with chlorine (or bromine) in the presence of sunlight and undergo substitution reaction. It is called photochemical reaction because it takes place in presence of sunlight.
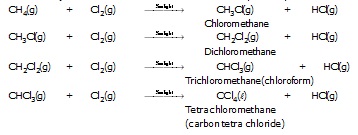
Q. Why is the conversion of ethanol to ethanoic acid an oxidation reaction? [NCERT]
Q. A mixture of oxygen and ethyne is burnt for welding. Can you tell why a mixture of ethyne and air is not used? [NCERT]
Ethanol is the second member of the homologous series of alcohols.
Preparation : By the fermentation of carbohydrates (sugar or starch).
Ethanol is prepared on commercial scale by fermentation of sugar. Fermentation is allowed to take place at 298 – 303 K
in the absence of air. This is ethanol (ethyl alcohol) gets oxidised to ethanoic acid
(acetic acid) in the presence of air.
![]()
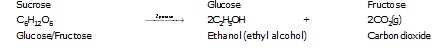
l Physical state / colour and odour : Pure ethanol is a colourless liquid having a pleasant smell and a burning taste.
l Boiling and Freezing points : It is a volatile liquid with a boiling point of 78.1°C, and freezing point is –118°C.
l Density : Ethanol is lighter than water as its density is 0.79 g ml–1 at 293 K.
l Solubility : Ethanol is miscible with water in all proportions, due to the formation of hydrogen bonds with water molecules.
l Conductivity : Ethanol is a covalent compound and does not ionise easily in water, hence it is a neutral compound.
l Action on Litmus : Ethanol is a neutral compound. So, it has no effect on the colour of litmus.
Þ Chemical properties of ethanol :
l Combustion (or burning) : Ethanol is highly inflammable liquid and readily burn in air with blue flame to form water vapour,
carbon dioxide and evolving heat. Thus, combustion of ethanol is an exothermic reaction.
Q. Carbon and its compounds are used as fuels. Give its main reason. [NCERT]
l Reaction with sodium metal : Ethanol reacts with sodium metal to produce sodium ethoxide and hydrogen gas is evolved.
![]()
Activity : To study the reaction of ethanol with sodium metal.
Materials : Ethanol, dry piece of sodium metal test tube.
* Take ethanol in a test tube.
* Add a dry piece of sodium metal.
* Bring a burning matchstick near the gas evolved to test it and record observation.
Observation : The gas burns in air with a pop sound which is the characteristics of hydrogen gas.
Conclusion : Alcohol react with sodium metal to liberate hydrogen gas.
* Reaction with ethanoic acid (Esterification reaction) :
The reaction in which an alcohol reacts with acetic acid in the presence of conc. H2SO4 to form an ester is called esterification.
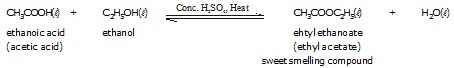
Note : Ester are sweet-smelling compounds and are used for making perfumes.
* Reaction with conc. sulphuric acid (Dehydration) :
Ethanol when heated with excess of concentrated sulphuric acid at 443 K, gets dehydrated to give ethene.
![]()
Note : The concentrated sulphuric acid can be regarded as a dehydrating agent which remove water from ethanol.
* Ethanol is present in alcoholic beverages such as beer, wine, whisky.
* As a solvent for paints, varnishes-dyes, cosmetics, perfumes, soaps and synthetic rubber etc.
* Ethanol is used in cough syrups, digestive syrups and tonics.
* A mixture of 80% rectified spirit and 20% petrol is called power alcohol. It is used as fuel in cars and aeroplanes.
* A mixture of ethanol and water has lower freezing point than water this mixture is known as antifreezing and is
used in radiators of vehicles in cold countries and at hill stations.
* As an antiseptic to sterilize wounds and syringes in hospitals.
* For the manufacture of terylene and polythene.
* As a preservative for biological specimens.
* Ethyl alcohol is used as hypnotic (induces-sleep).
Harmful effects of Alcohols :
* Consumption of small quantities of dilute ethanol causes drunkenness. Even though this practice is condemned,
it is a socially widespread practice. However, intake of even a small quantity of pure ethanol (called absolute alcohol) can be lethal.
Also long-term consumption of alcohol leads to many health problems.
* When large quantities of ethanol are consumed, it tends to slow metabolic processes and to depress the central nervous system.
This results in lack of coordination, mental confusion, drowsiness, lowering of normal inhibitions and finally stupor (unconscious state of wild)
* Drinking of alcohol over a long period of time damages liver.
Denatured Alcohol :
Ethanol to which certain poisonous and nauseating substances like methyl alcohol, pyridine etc. have been added is termed denatured alcohol.
Note : To prevent the misuse of ethanol (Alcohol), industrial alcohol is coloured blue so that it can be recognised easily.
Harmful effects of denatured alcohol :
* Methanol is highly poisonous compound for human beings. Methanol when taken, even in small amount, can cause death.
* Methanol gets oxidised to methanal in the liver, which causes coagulation of protoplasm.
* Methanol also effects the optic nerve and cause blindness.
* Ethanoic acid is commonly called acetic acid and belongs to the homologous series of carboxylic acid and is represented as CH3COOH.
* 5-8% solution of acetic acid in water is called vinegar and is used for preservating foods like sausage, pickles.
Physical properties :
* At ordinary temperature, ethanoic acid is a colourless liquid with a strong pungent smell and sour taste.
* Its boiling point is 391 K and its density at 273 K is 1.08 (heavier than water).
* It is miscible with water due to the formation of hydrogen bonds with water molecules.
* On cooling at 289.6 K, it turns in ice-like crystals, hence named as glacial acetic acid.
* It dissolves sulphur, iodine and many other organic compounds.
* It dimerise when dissolved in benzene.
* ![]()
Activity : To determine pH of acetic acid and hydrochloric acid.
Material : Acetic acid (1M), HCl (1M), blue litmus paper, universal indicator.
Procedure : Take two strips of blue litmus paper.
* Put a few drops of HCl on one of them and few drops of acetic acid on the other.
* Observe the change in colour.
* Take 1 ml of acetic acid in a test tube and add a few drops of universal indicator.
* Take 1 ml of HCl in a test tube and add few drops of universal indicator.
Observation : Both acetic acid and HCl turn blue litmus red showing that they are acidic in nature. pH of acetic acid and HCl are not equal.
Conclusion : HCl is strong acid than CH3COOH, therefore, pH of HCl will be lower than that of acetic acid.
* Reaction with alcohols (Esterification reaction) :
Ethanoic acid reacts with ethanol in the presence of conc. H2SO4 to form ethyl ethanoate which is an ester.
![]()
the reaction of carboxylic acid with an alcohol to form an ester is called "esterification".
Note : Ester can be hydrolysed in the presence of an acid or a base to give back the parent carboxylic acid and the alcohol.
Example :
(i) Ethyl ethanoate on acid hydrolysis gives ethanoic acid and ethanol.
![]()
(ii) Hydrolysis of ester in the presence of base (alkali) is called "Saponification reactions".
![]()
Note : Alkaline hydrolysis of higher esters is used in the manufacture of soaps.
Activity : To study the esterification process using ethanol and acetic acid.
Materials : Beaker, water, test tube, ethanol, acetic acid.
Conc. H2SO4 etc.
Procedure : Take 2ml of ethanol in a test tube.
* Add 2ml of ethanoic acid (acetic acid) in to it.
* Add few drops of conc. H2SO4.
* Warm it in a beaker containing water.
Observation : Pleasant fruity smelling compound
(called ester) is formed.
Conclusion : Acetic acid reacts with alcohol in
presence of conc. H2SO4
which act as a dehydrating agent to form ester.
Ethanoic acid decomposes sodium hydrogen carbonate and sodium carbonate with a rapid evoluation of carbon-dioxide gas.
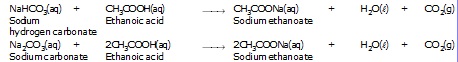
Note ; Reactions of ethanoic acid with NaOH, NaHCO3, Na2CO3 and active metals show that the hydrogen present in the carboxyl (–COOH) group is acidic in nature.
Activity : To study the reaction of carboxylic acid with sodium carbonate and sodium hydrogen carbonate.
Material : Ethanoic acid, Sodium carbonate, Sodium hydrogen carbonate.
Procedure :
* Take 1g of Na2CO3 and add 2ml of ethanoic acid into it.
* Pass the gas formed through lime water and note down the observation.
* Repeat the same procedure with sodium hydrogen carbonate and record observation.
Observation : Brisk effervescence due to carbon dioxide formed which turns lime water milky.
Conclusion : Acetic acid react with Na2CO3 and NaHCO3 to liberate CO2 gas.
Uses of ethanoic acid :
* Ethanoic acid is used in the manufacture of various dyes, perfumes and rayon.
* It is used for making vinegar.
* It is used for making white lead [2PbCO3 . Pb(OH)2] which is used in white paints.
* Its 5% solution is bactericidal (destroys bacteria).
* It is used in preparation of cellulose acetate which is used for making photographic film.
* It is used for coagulation of the latex.
* It is used for preparation of 2, 4-dichloro phenoxy ethanoic acid which is used as herbicide.
* Aluminium acetate and chromium acetate are used as mordants in dyeing and water proofing of fabrics.
Q. How would you distinguish experimentally between an alcohol and a carboxylic acid? [Ncert]
Q. What are oxidising agents? [Ncert]
Q. How can ethanol and ethanoic acid be differentiated on the basis of their physical and chemical properties? [Ncert]
Soap and detergents :
Soap and detergents are substances which are used for cleaning. There are two types of detergents :
1. Soap
2. Synthetic detergents
Soap : A soap is the sodium or potassium salt of a long-chain fatty acids (carboxylic acid or glycerol).
Activity :
Take about 10mL of water each in two test tubes.
Add a drop of oil (cooking oil) to both the test tubes and label them as A and B.
To test tube B, add a few drops of soap solution. Now shake both the test tubes vigourously for the same period of time.
Can you see the oil and water layers separately in both the test tubes immediately after you stop shaking them.
Leave the test tubes undisturbed for some time and observe. Does the oil layer separate out ? In which test tube does this happen first ?
this activity demonstrate effect of soap in cleasing as we know that most of the dirt is oily in nature and oil does it dissolve in water.
But know the question arise what are soap ? What are the detergent which one is more effect. How the work.
Soap is sodium or potassium salt a long change fatty acid (Carboxylic acid or Glycerol)
A soap has large non ionic hydrocarbon group and an ionic group. COONa.
Ex.of soap are :
(1) Sodium stearate (C17H35COONa)
(2) Sodium plamitate (C15H31COONa)
Soap are basic in nature so soap solution turn red litmus to blue.
Preparation of Soap :
The soap is prepared by heating animal fats or vegetable oils (olive oils, castor oil or palm oil)
with sodium hydroxide or potassium hydroxide.
The process of formation of soap by the hydrolysis of fat or oil with alkali is called saponification.
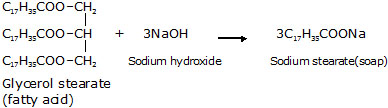
Oil or Fat + Sodium hydroxide ® Soap + glycerol
l A soap molecule contains two parts that interact differently with water, one part is a long hydrocarbon
(non-polar) chain, and other belongs to the –COONa group (Hydrophillic).
A soap molecule may be represented as :
The molecules of soap are sodium or potassium salts of long chain carboxylic acids. The ionic end of soap dissolves in water
while the carbon chain dissolves in oil. The soap molecules, thus form structures called micelles where one end of the molecules
is towards the oil droplet while the ionic end faces outside. This form an emulsion in water.
The soap micelle thus helps in dissolving the dirt in water and we can wash out clothes clean.
Q. Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also? [ncert]
Q. Explain the mechansim of the cleaning action of soaps. [ncert]
Activity : Take two clean test tubes and label them as 'A' and 'B'. Now put 10ml of hard water in each of the two test tubes.
Add five drops of soap solution in test tube 'A' and five drops of detergent solution in test tube 'B'. Shake the two test tubes
for the same period and observe if both the test tubes have the same amount of foam.
Find out in which of the two test tubes a curdy white mass is formed.
In which test tube do you get more foam ?
We get more foams in test tube .................... .
A white curdy precipitate is formed in test tube...............
Result (Conclusion) : Soaps are not effective in acidic medium.
When soaps is used for washing clothes with hard water, a large amount of soap is wasted in reacting with the calcium and
magnesium ions of hard water to form an insoluble precipitate called scum, before it can be used for the real purpose of washing soap.
A large amount of soap is needed for washing clothes. When the water is hard.
Activity :
Take two test tubes with a about 10 mL of hard water in each.
Add five drops of soap solution to one and five drops of detergent solution to the other.
Shake both test tubes for the same period
Do both test tubes have the same amount of foam ?
In which test tube is a curdy solid formed ?
Test tube in which ............................is present contain more amount of foam.
Curdy solids is form in the test tube containing ....................
Conclusion : Detergents have better cleasing action than soap. Detergents are generally ammonium or sulphonate salts of long chain carboxylic acids. The charged ends of these compounds do not form insoluble precipitates with the calcium and magnesium ions in hard water. Thus, they remain effective in hard water. Detergents are usually used to make shampoos and products for cleaning clothes.
Q. What change will you observe if you test soap with litmus paper (red and blue)? [NCERT]
Q. Would you be able to check if water is hard by using a detergents ?
Q. People use a variety to methods to wash clothes. Usually after adding the soap, they 'beat' the clothes on a stone, or beat it with a paddle, scrub with a brush or the mixture is agitated in a washing machine. Why is agitation necessary to get clean clothes ?
* The hydrocarbon chain is non-polar and water -hating (hydrophobic), while the other part is polar or water loving (hydrophilic).
* Hydrophilic part makes the soap soluble in water and hydrophobic part makes the soap insoluble.
* When soap is added to water, the soap molecules assume a configuration which increases the interaction of the water loving
heads with the water molecuels, and decreases the interaction between the water hating tails with the water molecules.
* The hydrophobic part of the soap molecules traps the dirt and the hydrophilic part makes the entire molecules soluble in water.
Thus, the dirt gets washed away with the soap.
* The water-hating, non polar tails clump together in a radial fashion with the water-loving, polar heads remaining at the
periphery of the clump, these clumps or droplets of soap molecules are called micelles.
Disadvantage of soap :
* Soaps are not effective in hard water : Hard water contains calcium ions (Ca2+) and magnesium ions (Mg2+).
These ions react with the carboxylate ions (RCOO–) of the soap forming an insoluble precipitate called scum. For example,
soap like sodium stearate (C17H35COONa) reacts with calcium and magnesium ions as per the following chemical equation.
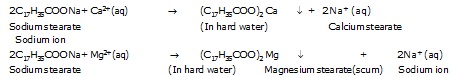
The scum gets attached to the clothes, utensils and even skin and thus, interferes with the cleansing ability of the
additional soap and makes the cleansing of clothes difficult. Moreover, large amount of soap is wasted in reacting with
calcium and magnesium ions present in hard water.
Q. Explain the formation of scum when hard water is treated with soap. [NCERT]
* Soaps are not effective in acidic medium : In presence of hydrogen ions (H+ ions), i.e. in acidic medium,
the carboxylate ions of soap (RCOO– ion) interact with hydrogen ions (H+) to form undissociated (free) fatty acid as represented below :
C17H35COO–(aq) + H+ ¾® C17H35COOH
carboxylate ion Carboxylic acid (Unionised)
As the fatty acids are weak acids, so they do not get ionised and hence, micelle formation is hindered, thus,
adversely affecting the cleansing property of soaps.
You will observe that the amount of foam in the two test tubes in different. The foam is formed to a greater extent in test tube 'B'
(containing detergent solution), while formation of a curdy white mass will be observed in test tube 'A'.
This activity clearly indicates that detergents can be used for cleansing purpose, even with hard water.
* Synthetic detergents are called soapless soap because they are not prepared from fatty acid and alkali.
* Synthetic detergents are sodium salts of sulphonic acids, i.e. detergents contain a sulphonic acid group
(–SO3H), instead of a carboxylic acid group (–COOH), on one end of the hydrocarbon chain.
* Synthetic detergents do not react with the ions present in hard water. Hence, synthetic detergents
have no problem in forming lather with hard water, i.e. their efficiency is not affected by hard water.
* Synthetic detergents can be used even in acidic solution and sea water, whereas soap cannot be used
in the acidic solution (due to precipitation of free acids)
* Synthetic detergents do not form insoluble salts of calcium or magnesium with hard water.
Hence, lesser amounts of synthetic detergents are required for washing.
Washing powder :
* Washing powders used for washing clothes contain only about 15 to 30 percent detergents by mass.
The remaining part is made of the following.
(i) Sodium sulphate and sodium silicate which keep the powder dry.
(ii) Sodium tripolyphosphate or sodium carbonate which maintains alkalinity for removing dirt.
(iii) Carboxymethylcellulose (CM-Cellulose) which keep the dirt particle suspended in water.
(iv) Sodium perborate (a mild bleaching agent) which impart whiteness to the materials (clothes, etc.) being washed.
• Carbon is versatile element that forms the basis of all living things.
• Carbon can form a vast variety of compounds because of its tetravalency and the property of catenation.
• Covalent bonds are formed between two similar or different atoms by sharing electrons in their valence shell,
such that both of them can achieve the structure of nearest noble gas.
• Carbon forms covalent bonds with itself as well as atoms of hydrogen, oxygen, nitrogen, sulphur and halogens.
• Carbon can form compounds having a straight chain between carbon atoms with a single bond, or double bond or triple bond.
It can also form compounds with branched chains and closed chains.
• logous series of carbon compounds is a group of carbon compounds having the same functional group with the same general formula.
• The functional groups such as alcohols, aldehydes, ketones, carboxylic acids and halogens impart characteristic properties to the carbon compounds.
• Carbon and its compounds are the major sources of fuels.
• Ethanol and ethanoic acid are most important compounds of carbon in our daily life.
• The soaps and detergents have cleansing action, because of the presence of hydrophobic and hydrophilic groups in their molecules,
which help in emulsifying oil, and hence, in the removal of dirt.
Q.1 What would be the electron dot structure of carbon dioxide which has the formula CO2?
Ans. The electron dot formula of CO2 is given below:
Q.2 What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur? [Hint: The eight atoms of sulphur are joined together in the form of a ring].
Ans. The electron dot structure of a molecule of sulphur which is made of eight atoms of sulphur is given below: Electron-dot structure of sulphur molecules, S8.
Q.3 How many structural isomers can you draw for pentane?
Ans. Three structural isomers can be drawn for pentane:
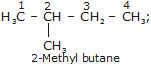
Q.4 What are the two properties of carbon which lead to the huge number of carbon compounds we see around us?
Ans. The two properties of carbon are its tetra covalency and the catenation which lead to huge number of carbon compounds we see around us.
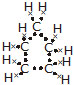
Q.5 What will be the formula and electron dot structure of cyclopentane?
Ans. The formula of cyclopentane will be C5H10. Its electron dot structure will be as shown below
Q.6 Draw the structures for the following compounds: (i) Ethanoic acid (ii) Bromopentane*
(iii) Butanone (iv) Hexanal. *Are structural isomers possible for bromopentane?
Ans. (i) Ethanoic acid (ii) Bromopentane
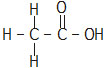
(iii) Butanone (iv) Hexanal
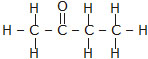
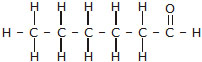
Bromopentane has a chain of five carbon atoms. It can exist in a number of forms which are structural isomers.
(i) (ii)
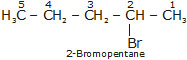
(iii) 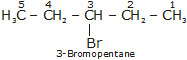
(v) 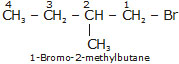
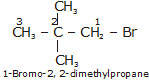
Q.7 How would you name the following compounds?
Ans. (i) (ii)
(iii)
Q.8 Why is the conversion of ethanol to ethanoic acid an oxidation reaction?
Ans. The conversion of ethanol (CH3CH2OH) to ethanoic acid (CH3COOH) is called an oxidation reaction
because oxygen is added to it during this conversion.
Q.9 The mixture of oxygen and ethyne is burnt for welding. Can you tell why a mixture of ethyne and air is not used?
Ans. A mixture of ethyne and air is not used for welding because burning of ethyne in air produces a sooty flame due
to incomplete combustion which is not enough to melt metals for welding.
When ethyne in burnt in oxygen, large quantity of heat alongwith light is produced. The heat evolved can be used
for gas welding which is usually carried to weld small broke pieces of articles made up of iron. Air mainly contains
a mixture of nitrogen (4 parts) and oxygen (1 part). As we know, nitrogen gas does not support combustion.
This means that in air, only oxygen will help in the combustion of ethyne. Therefore, it is always better to
use oxygen for the combustion of ethyne.
Q.10 How would you distinguish experimentally between an alcohol and a carboxylic acid?
Ans. Take the samples of alcohol and carboxylic acid in different test tubes and add sodium hydrogen carbonate in both the test tubes.
The test tube from which the effervescence evolves is carboxylic acid. The effervescence is due to the evolution of carbon dioxide.
CH3COOH + NaHCO3 ¾® CH3COONa + H2O + CO2
C2H5OH + NaHCO3 ¾® No evolution of CO2
Q.11 What are oxidising agents?
Ans. An oxidising agent is one which oxidizes other substances by providing oxygen or removing hydrogen.
The Commonly used oxidising agent are: Ozone, bromine water, a mixture of potassium dichromate and sulphuric acid or
a mixture of potassium permanganate and sulphuric acid.
Q.12 Would you be able to check if water is hard by using a detergent?
Ans. No, we cannot check the presence of hard water by using a detergent. Actually detergents produce foam in any type of water;
whether hard or soft. Therefore, a distinction between the two cannot be made. However, soaps can be used for this purpose.
Q.13 People use a variety of methods to wash clothes. Usually after adding the soap, they 'beat' the clothes on a stone,
or beat it with a paddle, scrub wit a brush or the mixture is agitated in a washing machine. Why is agitation necessary to get clean clothes?
Ans. The molecules of soap are sodium or potassium salts of long-chain carboxylic acids. The ionic-end of soap dissolves in water
while the carbon chain dissolves in oil. The soap molecules, thus form structures called micelles. When cloth is agitated,
the micelles containing oily or greasy dirt particles get removed from the surface of dirty cloth and into water and clothes gets cleaned.
Q.14 Ethane with the molecular formula C2H6 has
(A) 6 covalent bonds (B) 7 covalent bonds (C) 8 covalent bonds (D) 9 covalent bonds
Ans. (B) [Hint: The structure of C2H6 is]:
Q.15 Butanone is a four-carbon compound with the functional group
(A) carboxylic acid (B) aldehyde (C) ketone (D) alcohol
Ans. (C) [Hint: The structure of butanone is:]
Q.16 While cooking, if the bottom of the vessel is getting blackened on the outside it means that
(A) the food is not cooked completely
(B) the fuel is not burning completely
(C) the fuel is wet
(D) the fuel is burning completely
Ans. (B) The fuel is not burning completely.
Q.17 Explain the nature of the covalent bond using the bond formation in CH3Cl.
Ans. Covalent bond is formed by sharing the electrons between two atoms:
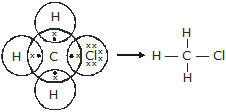
Q.18 Draw the electron dot structure for
(a) ethanoic acid (b) H2S (c) propanone (d) F2
Ans. The electron dot structures are given below:
(a) Ethanoic acid: 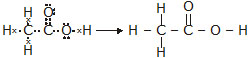
(b) Hydrogen sulphide:
(c) Propanone: 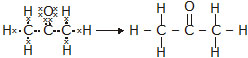
(d) Fluorine:
Q.19 What is an homologous series? Explain with an example.
Ans. A series of compounds in which the same functional group substitutes for hydrogen in a carbon chain is called homologous series.
These differ by –CH2 unit.
Exmple: Homologous series of alcohols: CH3OH, C2H5OH, C3H7OH. These differ by –CH2 unit.
Q.20 How can ethanol and ethanoic acid be differentiate on the basis of their physical and chemical properties?
Ans. Differentiation of ethanol and ethanoic acid on the basis of their physical properties. Ethanol has a pleasant odour whereas
Ethanoic acid smells like vinegar. Differentiation of ethanol and ethanoic acid on the basis of their chemical properties:
Take the samples of ethanol and ethanoic acid in different test tubes and add soldium hydrogen carbonate in both the test tubes.
The test tube from which the effervescence evolves is ethanoic acid. The effervescence is due to the evolution of carbon dioxide.
CH3COOH + NaHCO3 ¾® CH3COONa + H2O + CO2
C2H5OH + NaHCO3 ¾® No evolution of CO2
Q.21 Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?
Ans. Soap are sodium or potassium salts of long-chain carboxylic acids. The acid end of soap dissolves in water while the carbon chain dissolves in oil.
When soap is added to the water, the hydrophilic end (acid end) will align along the surface of water and the hydro
phobic tail (carbon chain) remains out of water. The cluster of
molecules in which hydrophobic tail are in the interior of cluster and the ionic ends are on the surface of cluster is called micelle.
The soap miceles thus helps in dissolving the dirt in water and we can wash our clothes clean. No, micelle will not be formed in their solvents such as ethanol.
Q.22 Why are carbon and its compounds used as fuels for most applications?
Ans. Carbon and its compounds are used as fuel because when they are burnt, they give large amount of energy in the form of heat.
Q.23 Explain the formation of scum when hard water is treated with soap.
Ans. Hard water contains soluble salts of sulphates and chlorides of calcium and magnesium. When soap is used to clean using hard
water calcium and magnesium present in hard water produce an insoluble substance called scum that remians after washing the cloth with water.
Q.24 What change will you observe if you test soap with litmus paper (red and blue)?
Ans. When we test soap with red and blue litmus paper red will turn blue but blue will remain as blue.
Q.25 What is hydrogenation? What is its industrial application?
Ans. Addition of hydrogen to an unsaturated hydrocarbon in presence of catalyst such as palladium or nickel to give saturated
hydrocarbons is called hydrogenation. The process is used to make healthy vegetable oils in industry.
Q.26 Which of the following hydrocarbons undergo addition reactions: C2H6, C3H8, C3H6, C2H2 and CH4.
Ans. Unsaturated hydrocarbons undergo addition reactions. The following are the unsaturated hydrocabons amongst the given examples: C3H6 and C2H2.
Q.27 Give a test that can be used to differentiate chemically between butter and cooking oil.
Ans. Take butter and cooking oil in two separate test tubes. Add bromine water to each test tube.
The one containing cooking oil will decolourise the light brown colour of bromine water, i.e., bromine water will become colourless.
The colour of Bromine water will remain as such in other test tube.
Q.28 Explain the mechanism of the cleaning action of soaps.
Ans. Cleaning action of soap has been explained with the help of figure. Soaps are moelcules in which the two ends have differing properties,
one is hydrophilic, that is it dissolves in water, while the other end is hydrophobic, that is it dissolves in hydrocarbons.
When soap is at the surface of water the hydrophobic 'tail' of soap will not be soluble in water and the soap will align
along the surface of water with the ionic end in water and the hydrocarbon 'tail' protruding out of water. Inside water,
these molecules have a unique orientation that keeps the hydrocarbon portion out of the water.
This is achieved by forming clusters of molecules in which the hydrophobic tails are in the interior of the cluster and the
ionic ends are on the surface of the cluster. This formation is called a micelle. Soap in the form of a micelle is able to clean,
since the oily will be collected in the centre of the micelle. The micelles are large enough to scatter light. Hence a soap solution appears cloudy.
Q.1 An organic compound 'A' has molecular formula C2H4O2 and is acidic in nature. On heating with alcohol and conc. sulphuric acid,
vapours with pleasant or fruity smell are given out. What is this chemical compound 'A' and what is the chemical equation involved in the reaction ? (CBSE All India 1999)
Ans. The compound 'A' with molecular formula C2H4O2 is ethanoic acid (acetic acid). Upon heating with ethanol
(ethyl alcohol) and concentrated sulphuric acid, ethyl ethanoate (ethyl acetate) is formed as the product. It is an ester with pleasant or fruity smell.
The reaction is known as esterification reaction.
Q.2 An organic compound 'A' is a constituent of antifreeze and has the molecular formula C2H6O. Upon reaction with alkaline KMnO4,
the compound 'A' is oxidised to another compound 'B' with formula C2H6O2. ldentify the compounds 'A' and 'B'.
Write the chemical equation for the reaction which leads to the formation of 'B'. (CBSE All India 2000 Comptt.)
Ans. The compound 'A' is ethanol and with alkaline KMnO4, it is oxidised to ethanoic acid 'B'. The chemical equation for the reaction is :
![]()
Q.3 Name the functional groups present in the following compounds :
(i) CH3—CH2—CH2—OH
(ii) CH3—CH2—CH2—COOH
(iii) CH3—CH2—CHO
(iv) CH3—CO—CH2—CH3
Ans. (i) —OH (ol)
(ii) —COON (oic acid)
(iii) —CHO (al)
(iv) —CO— (one)
Q.4 Write the formulae of the compounds and name the functional groups present in each of them
(i) Ethanoic acid
(ii) Propanone
(iii) Nitromethane (C.B.S.E. Delhi 2005)
Ans. (i) Ethanoic acid : CH3COOH (oic acid)
(ii) Propanone : CH3COCH3 (one)
(iii) Nitromethane : CH3NO2 (nitro)
Q.5 Name the enzyme which converts :
(i) milk into curd (yogurt)
(ii) cane sugar into glucose and fructose
(iii) glucose into ethanol. (C.B.S.E. Foreign 2005)
Ans. (i) Lactase converts milk into curd
(ii) Invertase converts cane sugar (sucrose) into glucose and fructose
(iii) Zymase converts glucose into ethanol.
Q.6 (i) Name the gas evolved during fermentation process. (C.B.S.E. Delhi 2006)
(ii) List the two products formed when enzyme invertase acts on sugar present in molasses.
Ans.(i) CO2 gas is evolved accompanied by brisk effervescence.
(ii) Glucose and fructose are the products when enzyme invertase acts on sucrose (Cl2H22O11) present in molasses.
Q.7 Complete the following equations and write the names of the products formed.
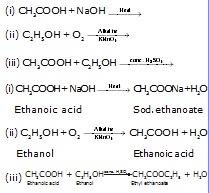
Q.8 Name the organic compound present in vinegar. Write a chemical equation which represents the commercial method
for the preparation of this compound from methanol. (C.B.S.E. All India 2007)
Ans. The organic compound present in vinegar is ethanoic acid also called acetic acid. For its commercial preparation.
Q.9 (a) Why does carbon form compounds by covalent bonding ?
(b) An organic acid 'X' is a liquid which often freezes during winter time in cold countries. It has the molecular formula C2H4O2.
On warming with ethanol in the presence of a few drops of sulphuric acid, a compound ‘Y’ with sweet smell is formed.
(i) Identify X and 'Y'.
(ii) Write chemical equation for the reaction involved. (C.B.S.E. Delhi 2008)
Ans. (a) Carbon forms a large number of organic compounds due to the self linking property known as catenation.
(b) The available information suggests that the organic acid X with molecular formula C2H4O2 is ethanoic acid (CH3COOH).
It reacts with ethanol in the presence of a few drops of sulphuric acid on warming to give ethyl ethanoate ester with a pleasant smell.
Q.10 Why do covalent compounds have low melting and boiling points ?
(C.B.S.E. All India 2008)
Ans. In covalent compounds, the atoms are linked by covalent bonds formed by electron sharing. Since no ions are present in these,
the attractive forces are quite weak. As a result, the covalent compounds have low melting and boiling points.
Q.11 (i) How are carboxylic acids different from mineral acids from ionisation point of view ?
(ii) Describe an activity to find how ethanoic acid reacts with sodium carbonate. Name the gas evolved. How can it be tested ? (C.B.S.E. All India 2008)
Ans. (i) Carboxylic acids (organic acids) are less ionised in solution as compared to mineral acids
(HCI, HNO3, H2SO4 etc.) Due to this reason, these are weaker acids than the mineral acids.
(ii) Take a small volume of ethanoic acid in a tube. Add a few drops of sodium carbonate (Na2CO3)
solution prepared in water to the tube. A colourless gas with brisk effervescence will evolve.
When the gas is passed through lime water, it will become milky.
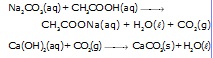
Q.12 (a) What is a functional group in a carbon compound ? Identify the functional group present in CH3COOH and C2H5OH.
(b) State the principle on which the cleansing action of soap is based.
(C.B.S.E. Foreign 2008)
Ans. (a) Functional group may be defined as an atom or group of atoms upon which the properties of a particular organic compound are based.
Different families differ in the functional groups.
Functional group in CH3COOH : (—COOH)
Functional group in C2H5OH : (—OH)
(b) The cleansing action of soap is based on its tendency to act as a bridge between water and oil drops containing dirt particles.
As a result, oil and water get mixed. They form a stable emulsion also called micelle. This helps in removing oil drops containing dirt particles from clothes. The clothes become clean.
Q.13 (a) Draw the structure of the following compounds
(i) Ethanoic acid
(ii) Butanone.
(b) Why is conversion of ethanol to ethanoic acid considered an oxidation reaction? (C.B.S.E. Forigen 2008)
Ans. (a)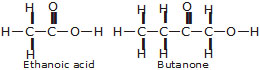
(b) When ethanol (C2H5OH) changes with ethanoic acid (CH3COOH)
• There is a decrease in the number of hydrogen atoms by two.
• There is an increase in the number of oxygen atoms by one. Therefore, the conversion represents an oxidation reaction.
Q.14 (a) What are esters ? How are they formed?
(b) Write two uses of esters ?
(CBSE Foreign 2008)
Ans. (a) Esters are the group of organic compounds which contain the function group (–COOR) called ester group.
The value of R may change as —CH3, —C2H5, —C3H7 etc. A few example of esters are :
Esters are formed as a result of chemical reaction called esterification.
Uses of esters
(i) Esters have pleasent smell. These are used as flavouring agents and also in perfumes.
(ii) Esters of glycerol known as triglycerides are used in the manufacture of soaps.This reaction is called saponification reaction.
Q.15 Distinguish between esterification and saponification reactions of organic compounds.
(C.B.S.E. All India 2008)
Ans. (a) In the esterification reaction an acid reacts with alcohol in the pressure of conc. H2SO4 to form an ester
with a pleasant or fruity smell. For example,
Saponification is quite different from esterification because in this case an ester reacts with an alkali
(NaOH or KOH) to form salt of acid and alcohol. For example,
C3COOC2H5 + NaOH CH3COONa + C2H5OH
Ethyl ethanoate Sod. ethanoate Ethanol
Q.16 (a) In organic compounds, which part largely determines the physical and chemical properties. (CBSE All India, 2008)
(b) Write chemical equation to represent the reaction of ethanol with acidified solution of potassium dicromate.
Ans. (a) In organic compounds, it is the functional group which largely determines the physical and chemical properties
of compounds. Actually, an organic compound is made up of two parts. These are alkyl group and the functional group.
Whereas the alkyl group remains the same (size may change) but the functional groups change.
These are responsible for the characteristics of the compounds. For example, the properties of alkanols
(–OH is the functional group) are different from those of alkanoic acid (–COON is the functional group).
For more details, consult text part.
(b) Acidified solution of potassium dichromate (K2O2O7) forms chromic acid (H2CrO4).
It releases oxygen to bring about the oxidation of ethanol first to ethanal and then to ethanoic acid.
Q.17 Give reason for the following:
(a) Air holes of the gas burners have to be adjusted when heated vessels get blackened by the flame.
(b) Use of synthetic detergents causes pollution problems. (C.B.S.E. Delhi, 2009)
Ans. (a) In case the vessel where cooking is done gets blackened from outside, this means that combustion is incomplete.
As a result, the carbon particles in the form of soot get deposited and the vessel becomes black from outside.
In order to check this, oxygen or air supply must be increased. This can be done only by adjusting the air holes of the gas burner.
(b) The pollution problems caused by the synthetic detergents is due to their non-biogradable nature.
These are actually long chain organic compounds which do not break or decompose in water. Naturally,
this will result in pollution problems. Some of the detergents are even of toxic nature and will make water unfit for drinking.
Q.1 Ethane, with the molecular formula C2H6 has
(A) 6 covalent bonds (NCERT)
(B) 7 covalent bonds
(C) 8 covalent bonds
(D) 9 covalent bonds
Q.2 Butanone is a four-carbon compound with the functional group (NCERT)
(A) Carboxylic acid
(B) aldehyde
(C) ketone
(D) alcohol
Q.3 While cooking, if the bottom of the vessel is getting blackened on the outside, it means that (NCERT)
(A) the food is not cooked completely
(B) the fuel is not burning completely
(C) the fuel is wet
(D) the fuel is burning completely
Q.4 A covalent bond is formed by
(A) complete transfer of electrons
(B) one sided sharing of electron
(C) mutual sharing of electron
(D) all of the three above.
Q.5 Which of the following compounds does not contain a multiple bond?
(A) Ethane
(B) Ethene
(C) Ethyne
(D) Benzene
Q.6 Which of the following is not a saturated hydrocarbon?
(A) Cyclohexane
(B) Benzene
(C) Butane
(D) Isobutane
Q.7 Benzene with molecular formula, C6H6, has
(A) 6 single bonds and 6 double bonds
(B) 12 single bonds and 3 double bonds
(C) 18 single bonds only
(D) 12 double bonds only
Q.8 The functional group in methanol and methanal respectively are :
(A) –OH, –CHO
(B) –CHO, –OH
(C) –OH, –COOH
(D) –CHO, –COOH
Q.9 Which of the following is not an allotropic form of carbon?
(A) Coal
(B) Fullerene
(C) Diamond
(D) Graphite
Q.10 Graphite is a soft lubricant extremely difficult to melt. The reason for this anomalous behaviour is that graphite
(A) has carbon atoms arranged in large plates of rings of strongly bound carbon atoms with weak interplate bonds
(B) is a non-crystalline substance
(C) is an allotropic form of carbon
(D) has only single bonds between carbon atoms
Q.11 Which of the following represent the correct order of unsaturation?
(A) Alkanes, alkenes, alkynes
(B) Alkanes, alkynes, alkenes
(C) Alkenes, alkynes, alkenes
(D) Alkynes, alkanes, alkenes
Q.12 The general formula of alcohol is
![]()
Q.13 Wine contains
(A) CH3OH
(B) C6H5OH
(C) C2H5OH
(D) CH3COOH
Q.14 The acid present in vinegar is
(A) CH3COOH
(B) HCOOH
(C) CH3CH2COOH
(D) CH3CH2CH2COOH
Q.15 The reaction 2C2H5OH + 2Na ¾¾® 2C2H5ONa + H2 suggests that ethanol is
(A) Acidic in nature
(B) Basic in nature
(C) Amphoteric
(D) Neutral
Q.16 Which of the following substance is added to denature ethanol?
(A) Methanol
(B) Benzene
(C) Copper nitrate
(D) Poision
Q.17 Which of the following substances cannot be used to distinguish ethanol from ethanoic acid?
(A) Na metal
(B) NaHCO3
(C) hot alkaline KMnO4 solution
(D) hot acidified K2Cr2O7 solution
Q.18 An example of soap is
(A) CH3COONa
(B) CH3ONa
(C) C17H35COONa
(D) C17H35COOC2H5
Q.19 Detergents are sodium or potassium salts of long chain
(A) aldehydes
(B) ketones
(C) carboxylic acids
(D) sulphonic acids
Q.20 Which of the following salts when dissolved in water produce hard water?
(A) Calcium sulphate
(B) Magnesium bicarbonate
(C) Calcium chloride
(D) Any of the above
Q.21 Which of the following represents Lewis structure of N2 molecule?
![]()
Q.22 Which of the following has the shortest carbon-carbon bond length?
(A) C2H2
(B) C2H4
(C) C2H6
(D) All have the same bond length
Q.23 Which of the following has the weakest carbon-carbon bond strength?
(A) C2H2
(B) C2H4
(C) C2H6
(D) All have the same bond length
Q.24 The hydrocarbon with the general formula CnH2n+2 is an -
(A) Alkane
(B) Alkene
(C) Alkyne
(D) unsaturated compounds
Q.25 Which of the following is an alkyne ?
(A) C6H6
(B) C6H12
(C) C6H10
(D) C6H14
Q.1 The ability of carbon atom to link with other carbon atom is known as ..............
Q.2 The hydrocarbons containing only single bonds are known as ..............
Q.3 Aliphatic hydrocarbons have been classified as alkanes, .............. and alkynes.
Q.4 .............. hydrocarbons undergo addition reaction.
Q.5 Isomers have same molecular formula but different .............. formula.
Q.6 Hydrocarbons are insoluble in ..............
Q.7 The next homologue of ethene is ..............
Q.8 During the formation of hydrogen molecule from its atoms, energy is ..............
Q.9 Bond between hydrogen and chlorine in HCl is ..............
Q.10 CH2 = CH2 + H2 CH3 – CH3
Q.11 The general formula of the homologous series of alcohols is ..............
Q.12 The functional group present in ethanol is ..............
Q.13 When ethanol is warmed with alkaline potassium permanganate, the product formed is ..............
Q.14 When a piece of sodium metal is added to ethanol, the gas formed with effervescence is ..............
Q.15 Ethene and ethyene are examples of .............. hydrocarbons.
Q.16 The ..............group in a soap molecule is hydrophilic.
C. True & false
Q.1 Methane belongs to the homologous series of alkanes.
Q.2 The compound having the molecular formula C4H10 does not show isomerism.
Q.3 The methane molecule has a pyrmidal shape.
Q.4 Methane undergoes substitution reaction.
Q.5 The consumption of ethanol increases the activity of the body.
Q.6 Ethanoic acid is used in the manufacture of textiles.
Q.7 Alkenes as well as alkynes decolourise bromine water.
Q.8 Vanaspati ghee is obtained by the hydrogenation of vegetable oil.
Q.9 Alkanes undergo substitution reactions.
Q.10 Alkenes and alkynes are unsaturated compounds.
Q.11 Ethanol is oxidised by alkaline KMnO4 to oxalic acid.
Q.12 Detergents give scum with hard water.
Q.13 The polar end in soap is called hydrophilic end.
Q.14 Methanol is safe to be used for drinking purpose.
Q.15 The reaction of ethanol with conc. H2SO4 gives ethane.
Q.16 Carboxylic acids react with alcohols to form esters.
Q.1 Name an allotrope of carbon which has 60 carbon atoms.
Q.2 What is combustion?
Q.3 What is the nature of substances that produce a flame?
Q.4 The molecular formula of a compound is C2H6O. Name its homologous series.
Q.5 What type of reactions are given by alkanes?
Q.6 What is the composition of natural gas used for cooking?
Q.7 Name an allotrope of carbon which contains both single and double bonds between carbon atoms.
Q.8 Write the name and molecular formula of alcohol derived from butane.
Q.9 What is rectified spirit?
Q.10 Give the name and structural formula of one homologue of HCOOH.
Q.11 Why does hard water not produce foam with soap easily?
Q.12 Name the hydrophobic and hydrophilic ends of a soap.
Q.13 An organic compound burns with a sooty flame. Is it saturated or unsaturated compound?
e. Short answer type question
Q.1 Name any four substance obtained from organic compounds which are used in our daily life.
Q.2 Hydrogen and chlorine react to form hydrochloric acid. Will they form an ionic or covalent bond?
Q.3 How many electrons a carbon atom shares with other carbon atom in the formation of acetylene molecule?
Q.4 Give examples of covalent compounds which contain :
(a) one unshared pair of electron
(b) two unshared pairs of electrons.
Q.6 What is meant by homologous series? Give its three important characteristics.
Q.7 Name the functional group present in :
(i) CH3CH2OH, and
(ii) HCOOH.
Q.8 The molecular formula of a hydrocarbon is C5H10. Name its homologous series.
Q.9 Write structural formulae of the following compounds.
(i) Ethyl alcohol
(ii) Acetaldehyde
(iii) Propionic acid
(iv) Butanone
Q.10 What is the IUPAC name of fourth member of alcohol series? Draw its two structural isomers.
Q.11 Write the general formulae of the following homologous series.
(i) Aldehydes
(ii) Ketones
(iii) Alkyne
Q.12 How do alcohols differ structrually from alkanes?
Q.13 Give the common and IUPAC names of the following alcohols :
(i) CH3OH
(ii) CH3CH2OH
(iii) CH3CHOHCH3
Q.14 Identify the functional group in CH3CH2OH. Give IUPAC name of the compound.
Q.15 Ethanol can be oxidised to ethanoic acid. Write the equation involved in the reaction.
Q.16 Name the oxidising agent which can oxidise:
(i) Ethanol to enthanal
(ii) Ethanol to ethanoic acid.
Q.1 What is covalent bond ? Draw electron dot structure for the following :
(i) Methane (ii) Carbon dioxide (iii) Ammonia (iv) Water
Q.2 Explain the following reaction with one example each :
(i) Substitution reactions
(ii) Addition reactions
(iii) Combustion reactions
(iv) Oxidation reactions
Q.3 (i) What are alcohols ? What is functional group?
(ii) Write names and formulae of first four members of alcohols family.
(iii) How does the second member of alcohol family react with
(a) Sodium metal
(b) Ethanoic acid
1. B 2. C 3. B 4. C
5. A 6. B 7. B 8. A
9. A 10. A 11. A 12. B
13. C 14. A 15. A 16. A
17. A 18. C 19. D 20. D
21. A 22. A 23. C 24. A
25. C
1. Catenation
2. Alkanes/saturated hydrocarbon
3. Alkenes 4. Unsaturated
5. Structural 6. Water
7. Propene 8. Released
9. Covalent 10. Ni catalyst
11. CnH2n+1OH. 12. Hydroxy
13. Ethanoic acid 14. Hydrogen
15. Unsaturated 16. Polar / COONa+
1. T 2. F 3. F 4. T
5. F 6. T 7. T 8. T
9. T 10. T 11. F 12. F
13. T 14. F 15. F 16. T
Q.1 Which of the following is an example of fossil fuel?
(A) coal gas
(B) coke
(C) natural gas
(D) producer gas
Q.2 Most of the fuels are:
(A) carbon compounds with sulphur
(B) nitrogen compounds with carbon
(C) carbon compounds with hydrogen
(D) none of these
Q.3 Destructive distillation of coal leads to the formation of
(A) wood
(B) kerosene
(C) ammoniacal liquor
(D) charcoal
Q.4 When stream is passed over hot coke, it produces
(A) producer gas
(B) synthesis gas
(C) tear gas
(D) coal gas
Q.5 Producer gas is a mixture of
(A) carbon monoxide and nitrogen gas
(B) carbon monoxide and hydrogen gas
(C) carbon monoxide and water vapour
(D) carbon monoxide and nitrous oxide
Q.6 Which of the following is not obtained on fractional distillation of petroleum?
(A) Paraffin wax
(B) Asphalt
(C) Coal gas
(D) Petroleum gas
Q.7 The property of an atom to form a bond with itself is known as:
(A) isomerism
(B) catenation
(C) allotropy
(D) none of these
Q.8 In the hydrocarbon HC º C – H, the covalency of carbon is:
(A) one
(B) two
(C) three
(D) four
Q.9 Which of the following are isomers?
(A) ethane and propane
(B) ethane and ethene
(C) ethene and ethyne
(D) butane and isobutane
Q.10 A hydrocarbon is:
(A) a compound containing carbon, hydrogen and oxygen
(B) is sodium hydrogen carbonate
(C) a compound containing carbon and hydrogen only
(D) a derivative of benzene
Q.11 The formula of ethyne is:
(A) C2H2
(B) C6H6
(C) C2H4
(D) C2H6
Q.12 The formula of tetrachloro methane is:
(A) CClH3
(B) CCl2
(C) CHCl3
(D) CCl4
Q.13 An unsaturated hydrocarbon
(A) contains six carbon atoms
(B) contains fewer hydrogen atoms that is needed
for carbon to have its usual valency of four
(C) contains excess hydrogen
(D) contains a chain of carbon atom
Q.14 Water gas consists of:
(A) water vapour and coal dust
(B) a mixture of carbon monoxide and hydrogen
(C) a mixture of carbon monoxide and nitrogen
(D) water vapour and methane
Q.15 The reaction of one mol of bromine with ethyne yields:
(A) BrCH2 – CH2Br
(B) BrCH = CHBr
(C) Br2CH – CHBr2
(D) CH3 – CH2Br
Q.16 Which of the following gas is knwon as illuminating gas?
(A) Ethane
(B) Ethene
(C) Ethyne
(D) Propane
Q.17 The gas used in the welding and cutting of metals is:
(A) ethane
(B) ethene
(C) ethyne
(D) propane
Q.18 Which of the following does not give a positive test
with alkaline solution of potassium permanganate?
(A) C10H22
(B) C6H12
(C) C10H18
(D) C10H20
Q.19 The process by which smaller molecules combine to
give a molecule with a higher molecular weight to known as:
(A) condensation
(B) polymerisation
(C) catenation
(D) isomerisation
1. C 2. C 3. A 4. B
5. A 6. C 7. B 8. D
9. D 10. C 11. A 12. D
13. B 14. B 15. B 16. C
17. C 18. A 19. B
1. Which of the following is a crystalline form of carbon?
(A) Charcoal
(B) Coal
(C) Graphite
(D) Lamp black
2. Which of the following is the purest form of carbon?
(A) Charcoal
(B) Coal
(C) Diamond
(D) Graphite
3. Each carbon atom in diamond is attached with four carbon atoms through four covalent bonds. This is why diamond is :
(A) A good conductor of heat
(B) A good conductor of electricity
(C) Purest form of carbon
(D) Such a hard substance
4. Which of the following statements regarding graphite is not correct?
(A) Graphite is a black and soft crystalline substance
(B) Graphite is manufactured by heating coke at 3000°C
(C) Graphite is a bad conductor of heat and electricity
(D) Graphite possesses a metallic lusture and soapy touch
5. Graphite has a much lower density than its allotrope diamond. It is because :
(A) Graphite has a characteristic greasy feel and metallic lusture
(B) Graphite has a more open structure than diamond
(C) Graphite is a good conductor of electricity while diamond is not
(D) Graphite is a good conductor of heat while diamond is not
6. Diamond and graphite resemble each other chemically because they :
(A) Are both allotropes of carbon and contains atoms of the same kind
(B) Have atoms arranged in the same way
(C) Are good conductors of heat and electricity
(D) Have high melting points
7. Animal charcoal, wood charcoal, lamp black and sugar charcoal are amorphours
forms of carbon. Which of the following contains calcium phosphate?
(A) Animal charcoal
(B) Wood charcoal
(C) Lamp black
(D) Sugar charcoal
8. A correct statement about charcoal, diamond, graphite and methane is that they all :
(A) Are allotropes of carbon
(B) Are soluble in water
(C) Form CO2 on combustion
(D) Have high melting points
9. The decolorising action of bone charcoal is due to its capacity to :
(A) Adsorb
(B) Float
(C) Oxidize
(D) Reduce
10. Which of the following statements is not correct?
(A) Graphite is much less dense than diamond
(B) Graphite is black and soft
(C) Graphite has low melting point
(D) Graphite feels smooth and slippery
11. Which of the following statements is not correct regarding graphite crystal?
(A) The carbon atoms are arranged in planar layers in the graphite crystals
(B) Within each layer of graphite, each carbon atom is bonded to three other
carbon atoms by covalent single bonds
(C) The atoms in each layer are not tightly bonded together
(D) The binding force between layers is weak allowing the layers to slip over each other
12. Which of the following statements correct?
(A) Graphite is a bad conductor of electricity
(B) Dry graphite in a vaccum is not slippery
(C) The adsorption of substance increases the friction as the layer slide past each other
(D) The graphite does not possess metallic properties
13. Which of the following statements regarding diamond is not correct?
(A) Diamond is clear and colourless substance
(B) Diamond is the hardest substance known
(C) Diamond is not brittle
(D) Diamond does not conduct electricity
14. Which of the following statements regarding diamond is not correct?
(A) Diamond does not refract light
(B) When proper cut and polished, a diamond reflects light in an array of many colours
(C) Diamond is less stable than graphite
(D) Impure sample of diamond is often black and are used in abrasives
15. Which of the following statements is not correct?
(A) Diamond can be made in a laboratory
(B) Gem-quality diamonds can be made in a laboratory
(C) Titanium dioxide and strontium titanate are the examples of diamond-like synthetic gems
(D) Graphite is the black ingredient in pencils
16. Organic compounds will always contain :
(A) Carbon
(B) Hydrogen
(C) Nitrogen
(D) Sulphur
17. The molecular shape of methane is :
(A) Octahedral
(B) Planar
(C) Pyramidal
(D) Tetrahedral
18. Methane, ethane and propane are said to form a homologous series because all are -
(A) Hydrocarbons
(B) Saturated compounds
(C) Aliphatic compounds
(D) Differ from each other by a CH2 group
19. The bond angle in methane molecule is :
(A) 90°
(B) 109°, 28°
(C) 120°
(D) 180°
20. Organic compounds are generally :
(A) Covalent in nature
(B) Ionic in nature
(C) Insoluble in orgainc solvents
(D) Soluble in water
21. Which of the following is not true for organic compounds?
(A) They form homologous series
(B) They show isomerism
(C) They are mostly obtained from vegetable or animal kingdom
(D) They are usually soluble in water but in-soluble in organic solvents
22. The isomers have the same
(A) Chemical properties
(B) Molecular formula
(C) Physical properties
(D) Structural formula
23. Which of the following pairs is an example of chain isomers?
(A) CH3CH2OH and CH3OCH3
(B) CH3CH2CHO and CH3COCH3
(C) CH3CH2CH2CH3 and
(D) Cl2C = CH2 and ClHC = CHCl
24. The number of chain isomers in pentane is :
(A) 1
(B) 2
(C) 3
(D) 4
25. The number of chain isomers in hexane is :
(A) 1
(B) 3
(C) 5
(D) 7
26. Carbon forms a large number of compounds because it :
(A) Has great chemical affinity
(B) Shows variable valency
(C) Forms many isotopes
(D) Has property of catenation
27. When methane is burnt in an excess of air, the products of combustion are :
(A) C and H2O
(B) CO and H2O
(C) CO2 and H2
(D) CO2 and H2O
28. The general formula of alkane series is :
![]()
29. Which of the following pairs of skelton in alkanes constitutes isomers?
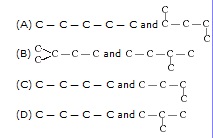
30. Which of the following gases is produced on heating 2 grams each of
sodium acetate and soda lime in a hard glass boiling tube?
(A) H2
(B) CO
(C) CO2
(D) CH4
31. Which of the following gases in called 'marsh gas'?
(A) H2
(B) CH4
(C) C2H4
(D) C2H2
32. The next higher homologue of propane is :
(A) C2H6
(B) C3H4
(C) C4H10
(D) C5H12
33. The IUPAC name of CH3CH2CH2CH3 is :
(A) Methylpropane
(B) Ethylethane
(C) Butane
(D) 1, 2-dimethylethane
34. The IUPAC name of is :
(A) 1, 1-methylethylpropane
(B) 2-ethylbutane
(C) 1-methyl-1-ethylpropane
(D) 3-methylpentane
35. Which of the following gases is liberated when a mixture of sodium propanoate and soda lime is heated?
(A) CH4
(B) CH3CH3
(C) CO2
(D) CH3CH2CH3
36. The final product of chlorination of methane in the sun light is :
(A) CH3Cl
(B) CH2Cl
(C) CHCl3
(D) CCl4
37. Which of the following types of reactions is characteristic of compounds having a double bond?
(A) Addition
(B) Condensation
(C) Isomerization
(D) Substitution
38. Alkenes are characterize by :
(A) C – C bonds
(B) C = C bonds
(C) C º C bonds
(D) Cyclic structure
39. Which of the following gases decolorises bromine water?
(A) CH4
(B) C2H2
(C) C2H6
(D) C3H8
40. Which of the following gases decolorises alkaline KMnO4 solution?
(A) CH4
(B) C2H2
(C) C2H6
(D) C3H8
41. The IUPAC name of CH3CH2CH = CH2 is :
(A) Butene
(B) Isobutene
(C) Butene-2
(D) 3-methylpropene
42. The IUPAC name of = CH CH3 is :
(A) Butene
(B) Isobutene
(C) But-2-ene
(D) 3-methylprop-2-ene
43. The treatment of ethene with alkaline solution of KMnO4 produces :
(A) CH3CH2OH
(B) HOCH2CH2OH
(C) CH3CHO
(D) CH3COCH3
44. The IUPAC name of acetylene is :
(A) Ethane
(B) Ethene
(C) Ethylene
(D) Ethyne
45. In acetylene, the two carbon atoms are joined by a :
(A) Single bond
(B) Double bond
(C) Triple bond
(D) Ionic bond
46. The general formula of alkyne is :
(A) CnHn
(B) CnH2n – 2
(C) CnH2n
(D) CnH2n + 2
47. Alkynes have in their molecule :
(A) Four hydrogen atoms more than in a molecule of corresponding alkane
(B) Two hydrogen atoms more than in a molecule of corresponding alkane
(C) Two hydrogen atoms less than in a molecule of corresponding alkane
(D) Two hydrogen atoms less than in a molecule of corresponding alkene
48. A compound with the molecular formula C2H2 must contain :
(A) All single bonds
(B) One double bond
(C) One triple bond
(D) None of the above
49. When ethyne is burnt in excess of air, the products of combustion are :
(A) C and H2O
(B) CO and H2O
(C) CO2 and H2
(D) CO2 and H2O
50. Which gas is produced when water reacts with calcium carbide?
(A) CH4
(B) C2H2
(C) C2H4
(D) C3H8
51. Which of the following gases burns with a sooty flame?
(A) CH4
(B) C2H2
(C) C2H6
(D) C3H8 < C2H6
52. The IUPAC name of is :
(A) Pentyne
(B) 4-methylbutyne
(C) 1-methylbute-3-yne
(D) Propylethyne
53. Which of the following gases is produced when cold water is added on calcium carbide?
(A) C2H2
(B) C2H4
(C) C2H6
(D) C3H8
54. Across a triple bond, the number of molecules of Br2 that can be added is :
(A) One
(B) Two
(C) Three
(D) Zero
55. When acetylene is passed over heated iron tube, the product obtained is :
(A) C2H2
(B) C4H4
(C) C6H6
(D) C8H8
56. The treatment of acetylene with alkaline KMnO4 solution produces :
(A) CH3CHO
(B) HOCH2CH2OH
(C)
(D)
57. The gas used in the flame produced for welding metals is :
(A) Methane
(B) Methene
(C) Methyne
(D) Ethyne
58. The amount of oxygen used in the combustion of 1 mol of ethene is :
(A) 1 mol
(B) 2 mol
(C) 2.5 mol
(D) 3 mol
59. The amount of oxygen used in the combustion of 1 mol of ethene is :
(A) 1 mol
(B) 2 mol
(C) 2.5 mol
(D) 3 mol
60. The treatment of ethanol with concentrated sulphuric acid produces :
(A) Ethane
(B) Ethene
(C) Ethyne
(D) None of these
61. The general formula of alcohols is :
(A) CnH2nOH
(B) CnH2n+1OH
(C) CnH2n+2OH
(D) CnH2n–1OH
62. The IUPAC name of is :
(A) 3-methylethanol
(B) Propanol
(C) 1-hydroxypropane
(D) 1-hydroxy-2-ethane
63. The number of oxygen molecules used in the combustion
of 1 molecule of ethanol is :
(A) 1
(B) 2
(C) 3
(D) 4
64. The final product obtained in the oxidation of ethanol by acidic potassium dichromate is :
(A) C2H5OH
(B) CH3CHO
(C) CH3COOH
(D) CH4COOH
65. The distillation of a mixture of ethanol and bleaching powder produces :
(A) Ethanal
(B) Chloral
(C) Chloroform
(D) Carbon tetrachloride
66. The treatement of ethanol with alkaline iodine solution produces :
(A) Ethanal
(B) Iodorom
(C) Carbon tetraiodide
(D) Diiodomethane
67. Which of the following gases gives a reddish brown precipitate with
ammoniacal cuprous chloride solution?
(A) CH4
(B) C2H2
(C) C2H4
(D) C2H5OH vapours
68. 'Drinking alcohol' is very harmful and it ruins the health. 'Drinking alcohol' stands for :
(A) Drinking methyl alcohol
(B) Drinking ethyl alcohol
(C) Drinking propyl alcohol
(D) Drinking isopropyl alcohol
69. Sodium is used for testing an :
(A) Alcohol
(B) Aldehyde
(C) Alkane
(D) Alkene
70. An alcohol produces a characteristic colour when treated with
ceric ammonium nitrate solution. The colour is :
(A) Blue
(B) Green
(C) Yellow
(D) Red
71. The general formula of a carboxylic acid is :
(A) CnH2n–1COOH
(B) CnH2nCOOH
(C) CnH2n+1COOH
(D) CnH2n+2COOH
72. The IUPAC name of is :
(A) Propionic acid
(B) Propanoic acid
(C) 1-methylethanoic acid
(D) 2-methylethanoic acid
73. The treatment of acetic acid with lithium aluminium hydride produces :
(A) Methanol
(B) Ethanol
(C) Ethanal
(D) Methanal
74. The treatment of CH3COOH with C2H5OH in the pressence of
concentrated sulphuric acid produces :
(A) CH3COOCH3
(B) CH3COOC2H5
(C) C2H5COOCH3
(D) C2H5COOC2H5
75. Which of the following compounds gives effervescence with sodium bicarbonate?
(A) HCHO
(B) CH3CHO
(C) CH3COOH
(D) CH3COOC2H5
77. Vinegar is a dilute solution of
(A) Acetic acid
(B) Propanoic acid
(C) Butanoic acid
(D) Formaldehyde
78. The general formula of an aldehyde is :
(A) CnH2nCHO
(B) CnH2n–1CHO
(C) CnH2n+2CHO
(D) CnH2nO
79. Formaline is :
(A) 40% aqueous solution of formaldehyde
(B) 20% aqueous solution of formaldehyde
(C) 40% solution of formaldehyde in ethanol
(D) 40% solution of formaldehyde in chloroform
80. The starting substances used in bakelite resin is :
(A) HCHO + C6H5OH
(B) CH3CHO + C6H5CHO
(C) HCHO + C6H5CHO
(D) HCHO + CH3CHO
81. Which of the following compounds deposits shining silver with Tollens reagent (ammoniacal silver nitrate solution)?
(A)CH3CHO
(B) CH3OH
(C) CH3COOH
(D) CH3CH2OH
83. Which of the following substances will forms red precipitate with Fehling's solution?
(A) CH3CH2OH
(B) CH3COCH3
(C) CH3COOH
(D) CH3CHO
84. Tollens reagent is :
(A) Ammoniacal solution of copper sulphate
(B) Ammoniacal solution of silver nitrate
(C) Ammoniacal solution of ferrous sulphate
(D) Ammoniacal solution of sodium chloride
85. The IUPAC name of is :
(A) Propanal
(B) Propanol
(C) Propanoic acid
(D) 2-methylethanal
86. The oxidation of CH3CH2CHO produces :
(A) CH3COOH
(B) CH3CH2COOH
(C) CH3COCH3 (D)
87. Which of the following compounds is phenol?
(A) C6H5COOH
(B) C6H5CHO
(C) C6H5OH
(D) C6H5CH2OH
88. Identify the 'product' in the reaction CH3COOH + C2H3OH 'product' + H2O
(A) Alcohol
(B) Aldehyde
(C) Ketone
(D) Ester
89. Identify the 'product' in the reaction HCHO + Ag2O 'product' + 2Ag
(A) Alcohol
(B) Acid
(C) Ether
(D) Ester
90. Identify the product in the reaction HCHO + 2CuO 'product' + Cu2O :
(A) Alcohol
(B) Acid
(C) Ether
(D) Ester
91. Identify the product in the reaction CH º CH + Br2 CHBr = CHBr 'product' :
(A) CH2BrCH2Br
(B) CHBr2CHBr2
(C) CH3CHBr2
(D) CH3CBr3
92. Identify the 'product' in the reaction CH3CH2OH 'product' + H2O :
(A) Acid
(B) Aldehyde
(C) Ketone
(D) Ester
93. Identify the 'product' in the reaction CH3CH2COONa 'product' + Na2CO3
(A) Alkane
(B) Alkene
(C) Alkyne
(D) Acid
94. Identify the 'product' in the reaction CH3COOH 'product' + H2O :
(A) Aldehyde
(B) Alcohol
(C) Ketone
(D) Acid
95. The fruity smell is of a/an :
(A) Aldehyde
(B) Ketone
(C) Alcohol
(D) Ester
96. The 'product' in the reaction CH3CH2OH 'product' + H2O is :
(A) Alkane
(B) Alkene
(C) Alkyne
(D) None of these
97. The 'product' in the reaction Glucose 'product' + 2CO2
(A) Methanol
(B) Ethanol
(C) Methane
(D) Water
98. The fermentation reactions are carried out in temperature range of :
(A) 20–30 °C
(B) 30–40 °C
(C) 40–50 °C
(D) 50–60 °C
99. The 'product' of the reaction CH2 = CH2 'product' is :
(A) CH3COOH
(B) HOCH2CH2OH
(C)
(D) None of these
100. The product of CH2 = CH2 + Cl2 'product' is :
(A) CH3CHCl2
(B) CH2 = CHCl
(C) CH2 = CCl2
(D) ClCH2CH2Cl
101. Octane number is zero for
(A)n-heptane
(B) 2, 3, 3-trimethylpentane
(C) 2, 2, 4-trimethylpentane
(D) n-octane
102. Octane number is 100 for
(A) p-pentane
(B) 2, 3, 3-trimethylpentane
(C) 2, 2, 4-trimethylpentane
(D) n-octane